Limiting Reactants
In a perfect world, we have just the right amount of ingredients for a reaction, known as being balanced. However, in reality, one reactant often limits the amount of product formed. This is where the concept of a limiting reactant comes into play, defining the maximum product yield. To identify the limiting reactant, follow these steps: write a balanced equation, convert each reactant to the mass of desired product, and determine which reactant produces the least. Additionally, methods exist to calculate the excess amounts of any unreacted reactants.

Limiting Reactants
E N D
Presentation Transcript
In a perfect world… • We have the perfect amount of out “ingredients” to make our desired product. • This is known as being balanced. • 2 Na + Cl₂ 2 NaCl
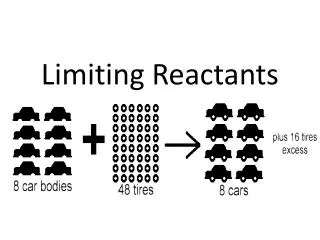
In the REAL world • Nothing is perfect… • One of the reactants (ingredients) limits how much product we can make. • Limiting Reactant- the reactant that determines how much product we can make. • 10 buns, 20 patties, 2 slices of cheese, and 40 strips of bacon 1 bacon double cheeseburger
How do I know the limiting reactant? Step 1: write out balanced equation. Step 2: convert each reactant to mass of product Step 3: the reactant that makes the LEAST amount of product is the limiting reactant.
What mass of Ag₂S is produced from a mixture of 2.0 g Ag and 2.0 g S ? Step 1: 16 Ag + S₈ 8Ag₂S
How do I find out how much reactant is left/ in excess ? • Step 1: Convert limiting reactant into mass of non-limiting reactant • Step 2: Subtract amount used from the amount at the beginning.