II. Limiting Reactants
Stoichiometry – 3.7. II. Limiting Reactants. A. Limiting Reactants. Available Ingredients 4 slices of bread 1 jar of peanut butter 1/2 jar of jelly. Limiting Reactant bread. Excess Reactants peanut butter and jelly. A. Limiting Reactants.

II. Limiting Reactants
E N D
Presentation Transcript
Stoichiometry – 3.7 II. Limiting Reactants
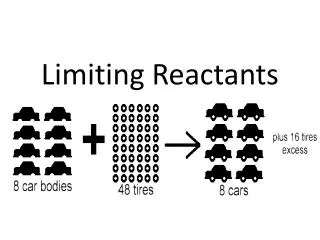
A. Limiting Reactants • Available Ingredients • 4 slices of bread • 1 jar of peanut butter • 1/2 jar of jelly • Limiting Reactant • bread • Excess Reactants • peanut butter and jelly
A. Limiting Reactants • In a laboratory, usually one or more of the reactants are present in excess. There is more than the exact amount required to react • Once one of the reactants is used up, no more product can form
A. Limiting Reactants • Limiting Reactant • used up in a reaction • Limits the amount of reactant that can combine and determines amount of product • determines the amount of product that can form • Excess Reactant • added to ensure that the other reactant is completely used up • cheaper & easier to recycle
B. Limiting Reactants 1. Write a balanced equation. 2. For each reactant, calculate the amount of product formed. 3. Smaller answer indicates: • limiting reactant • amount of product
B. Limiting Reactants • 79.1 g of zinc react with 0.90 L of 2.5M HCl. Identify the limiting and excess reactants. How many liters of hydrogen are formed at STP? Zn + 2HCl ZnCl2 + H2 ? L 79.1 g 0.90 L 2.5M
B. Limiting Reactants Zn + 2HCl ZnCl2 + H2 ? L 79.1 g 0.90 L 2.5M 79.1 g Zn 1 mol Zn 65.39 g Zn 1 mol H2 1 mol Zn 22.4 L H2 1 mol H2 = 27.1 L H2
B. Limiting Reactants Zn + 2HCl ZnCl2 + H2 ? L 79.1 g 0.90 L 2.5M 0.90 L 2.5 mol HCl 1 L 1 mol H2 2 mol HCl 22.4 L H2 1 mol H2 = 25 L H2
B. Limiting Reactants left over zinc Zn: 27.1 L H2 HCl: 25 L H2 Limiting reactant: HCl Excess reactant: Zn Product Formed: 25 L H2
LIMITING REACTANT • Try Example Problem #2 • Method 1: Convert both reactants to product. See which is less. • Method 2: Convert one reactant to another. See how much is needed.
LIMITING REACTANT • Problem #2: • HF: limiting • 4.0 mol excess SiO2
C. Percent Yield 1. actual yield: measured amount of product obtained from a reaction; measured in actual lab; less than theoretical yield due to experimental errors 2. theoretical yield: maximum amt. of product that could ideally be obtained from a given amount of reactant
C. Percent Yield measured in lab calculated w/stoich.
C. Percent Yield • When 45.8 g of K2CO3 react with excess HCl, 46.3 g of KCl are formed. Calculate the theoretical and % yields of KCl. K2CO3 + 2HCl 2KCl + H2O + CO2 45.8 g ? g actual: 46.3 g
C. Percent Yield K2CO3 + 2HCl 2KCl + H2O + CO2 Theoretical Yield: 45.8 g ? g actual: 46.3 g 45.8 g K2CO3 1 mol K2CO3 138.21 g K2CO3 2 mol KCl 1 mol K2CO3 74.55 g KCl 1 mol KCl = 49.4 g KCl
C. Percent Yield 46.3 g 49.4 g K2CO3 + 2HCl 2KCl + H2O + CO2 Theoretical Yield = 49.4 g KCl 45.8 g 49.4 g actual: 46.3 g 100 = 93.7% % Yield =