Liquids Solids
Heat of Fusion/Vaporization. H2O(s) ----> H2O(l) ?Hfusion = 6.02 kj/molH2O(l) ----> H2O(g) ? Hvaporization = 40.6 kj/molFrom the ? Ho values above, which two states are most similar?How do the attractive forces between the molecules compare in these two states to the third state? . Liquids

Liquids Solids
E N D
Presentation Transcript
1. Liquids & Solids
Chapter 13
2. Heat of Fusion/Vaporization H2O(s) ----> H2O(l) ?Hfusion = 6.02 kj/mol
H2O(l) ----> H2O(g) ? Hvaporization = 40.6 kj/mol
From the ? Ho values above, which two states are most similar?
How do the attractive forces between the molecules compare in these two states to the third state?
3. Liquids & Solids The liquid and solid states are considered to be the condensed states.
Liquids & solids have much higher densities than gases and are not compressible.
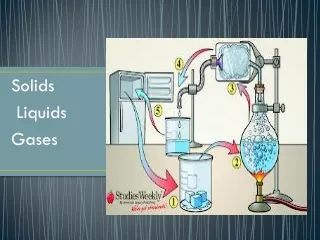
4. Three States of Matter