Polymers
Polymers. Polymers are giant molecules that are made up of many, many smaller molecules. Building blocks for polymers are called monomers . Examples: plastics, rubber etc. Biopolymers: proteins, polysaccharides , nucleic acids. Polymers. Biopolymers:. Proteins.

Polymers
E N D
Presentation Transcript
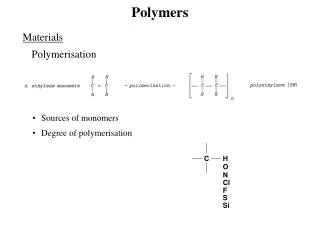
Polymers Polymers are giant molecules that are made up of many, many smaller molecules. Building blocks for polymers are called monomers. Examples: plastics, rubber etc. Biopolymers: proteins, polysaccharides, nucleic acids
Polymers Biopolymers: Proteins • Basic building blocks are amino acids • Amino acids linked together into proteins by amide groups • Peptide bonds formed by condensation reactions between two • amino acids. e.g. Alanine + Glycine: Ala Gly Gly-Ala
Polymers Biopolymers:
Polymers Amino acids:
Polymers Polysaccharides (Carbohydrates) • Monosaccharides linked together by ether-bridges Representative segment of cellulose: a tough fibre.
Polymers Nucleic acids Composed of: H3PO4 molecule Organic base 5-C sugar
Polymers In order to get a polymer formed we need a bifunctional molecule.
Polymers Addition Polymerization Example: ethylene H2C=CH2, can polymerize by opening the C–C bond to form C–C bonds with adjacent ethylene molecules (with the help of radicals). The result: polyethylene. This is called addition polymerization because ethylene molecules are added toeach other.
Polymers Step 1Initiation:generation of radicals from catalyst Step 2 radical adds to ethylene and polymerisation starts
Polymers This is the repeat unit
Polymers repeat unit
Polymers Condensation Polymerization Condensation Polymerization: molecules are joined by the elimination of a small molecule (e.g. water): Example of condensation polymerization: formation of nylon.
Polymers Another example of a condensation polymerisation is the formation of nylon 6. Used for clothing and mountaineering ropes amongst others
Polymers Types of Polymers Plastic Materials that can be formed into shapes by application of heat and pressure. Thermoplastics: • Hard at room temp. • Become soft + viscous when heated. can be • shaped more than once. • Little or no cross-linking individual chains can slip • past each other. • E.g.’s include polyethylene, nylon, polystyrene….
Polymers Thermosetting resins: • Become highly cross-linked when heated. solidify • into a hard, insoluble mass. • Can only be shaped once, polymerisation irreversible. • Can withstand high temperatures. • For example, Bakelite; used for adhesives, moulded • parts and coatings: Phenol formaldehyde
Polymers Elastomers: • Have the ability to stretch out & spring back to their • original shapes. • Have a modest amount of cross-linking. • Polymer chains have irregular shapes. • Most common example is natural rubber
Polymers Structure and Physical Properties of Polymers Polymer chains tend to be flexible and easily entangled or folded due to free rotation around the C–C single bonds. Some regions of the polymer, may however, display a more ordered arrangement of chains than other regions:
Polymers Structure and Physical Properties of Polymers The degree of crystallinity is a measure of the extent of such ordering. More ordering in a polymer = denser, harder, less soluble polymers that are more resistant to heat. e.g. Properties of PE as a Function of Crystallinity.
Polymers • Structure and Physical Properties of Polymers • Stretching or extruding a polymer can increase crystallinity. • Degree of crystallinity is also determined by average molecular mass: • Low density polyethylene (LDPE) has an average molecular mass of 104 amu (used in plastic wrap); • High density polyethylene (HDPE) has an averagemolecular mass of 106 amu (used in milk cartons).
Polymers • Structure and Physical Properties of Polymers LDPE: • Obtained by polymerization of ethylene at high pressure • and high temperature. • Polymer chains have irregular branches and cannot pack • together in an ordered way. • Result: LDPE is an open polymer of low density and little • mechanical strength.
Polymers • Structure and Physical Properties of Polymers HDPE: • Polythene can also be prepared catalytically at lower • pressures and temperatures. • Result: regular non-branched chain polymer which is highly • ordered or crystalline. • HDPE is tough and strong and the ordered structure means • that it has higher density.
Polymers • Structure and Physical Properties of Polymers Polyethylene is a very versatile material & it’s properties can be “fine tuned” by varying:
Polymers • Cross-Linking Polymers • Bonds formed between polymer chains make the polymer stiffer. • Natural rubber is too soft and chemically reactive to make a useful material. • By vulcanizing the rubber (cross-linking the polymer chains) useful materials are made. • Rubber is usually cross-linked with sulfur. • Cross-linked rubber is stiffer, more elastic and less susceptible to chemical reaction.
Polymers Cross-Linking Polymers