Polymers
Polymers. Polymers . C.X.C objectives. Students should be able to : Define polymers Distinguish between addition and condensation as reactions in the formation of polymers Name examples of polymers formed by: ( i ) addition reactions (ii)condensation reactions

Polymers
E N D
Presentation Transcript
Polymers Polymers
C.X.C objectives • Students should be able to : • Define polymers • Distinguish between addition and condensation as reactions in the formation of polymers • Name examples of polymers formed by: (i) addition reactions (ii)condensation reactions • Draw diagrams to represent the formulae of monomers • State at least one use of each of the following types of polymers : (i) polyalkene (ii) polyamide (iii) polyester (iv) polysaccharide • Show how the monomers are linked in the structure of a polymer • Demonstrate the differences in properties between a monomer and the polymer it forms.
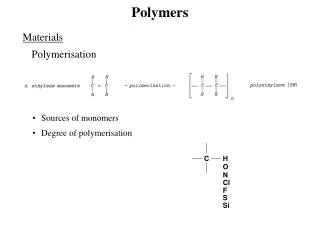
Polymers • Obj 1. Students should be able to define polymers : • What is a polymer ? • Polymers are macromolecules formed by linking together thousands of small molecules called monomers, usually in chains. Polymers are formed by polymerisation. • Some polymers occur naturally whereas some are man-made (synthetic). Synthetic polymers are referred to as Plastics but will be further discussed later in this module.
Polymerisation • Obj. 2. Distinguish between addition and condensation as reactions in the formation of polymers. • Polymerisation is the process whereby a polymer is formed from monomers. This can happen in two ways. Addition Polymerisation Condensation Polymerisation Occurs when monomers join with the elimination of a small molecule e.g. water from between each unit. Occurs when unsaturated monomers are linked to form a saturated polymer.
Addition Polymerisation • An addition polymer is constructed of one type of monomer. This is an unsaturated molecule (usually an alkene C C ) • Addition polymers are referred to as Polyalkenes. • The polymer is formed when the double bond breaks and the units join together . • Only one type of product is formed.
Naming Polymers • To name a polymer, the prefix ‘Poly’ is placed before the name of the monomer. • For example: Polypropene Polystyrene Polyethene
Addition Polymers • Polyalkenes have properties of substances commonly named PLASTICS. Plastics are synthetic polymers.
Condensation Polymerisation • Condensation polymerisation describes a process whereby the polymer is formed when monomer units join together with the elimination of a small molecule, usually BUT NOT ALWAYS water. (HCl or NH3 could be eliminated as well.) • In order for monomers to form condensation polymers, the monomer must have two active sites ( point at which the monomers join). • Two products are formed as a result of this type of polymerisation. • It is important to note that a condensation polymer can have monomers of one or two types.
Condensation Polymerisation • There are two types of condensation polymerisation. Natural Condensation polymers e.g protein and starch Synthetic or man-made condensation polymers e.g. nylon and terylene
Types of linkage • Condensation polymers can be divided into groups based on the type of linkage between the monomer units. • Polyamides – Amide Linkage • Polyesters- Ester Linkage C O Polysaccarides- Saccaride Linkage O
Types of condensation polymers • Polyamides • Polyesters • Polysaccharides
Polyamides • Protein is a natural polyamide. The monomers which make up proteins are amino acids.
Polyamides cont’d : • Two amino acids join together to form a dipeptide. A ‘H’ from the amine ( NH2) group of one of the amino acids and an ‘OH’ from the other amino acid condense to form water and join the two monomers.
Polyamides cont’d: • Nylon is an example of a synthetic polymer. Nylon is special in that it is formed by two different monomers, a diacid and a diamine.
Polyesters • Polyesters are synthetic fibres, such as terylene, made as imitations of natural materials like wool and cotton. • Polyester structure consist of many monomers joined together by ester bonds. • The monomers in polyester are : diacid + dialcohol
Polysaccarides • Polysaccarides are natural polymers such as starch and cellulose. The monomers are monosaccaridese.g. fructose or glucose (simple reducing sugars) • Two glucose units join together by the elimination of water to produce a disaccaride (sucrose) and many glucose units join together to form the polysaccaride- starch. • Starch- [ O X O X O ]n
Hydrolysis • Hydrolysis of Polymers- • The word ‘hydrolysis’ means to split up by the addition of water. Hydro- water, lysis-to break up/separate. • Polymers which undergo hydrolysis are broken up into their respective monomers when water is added.
Hydrolysis • Carbohydrates and Proteins can be hydrolysed in two ways. • 1) In the body during digestion by enzymes. • 2) In the lab, by boiling with dilute Hydrochloric acid or sulphuric acid.
Properties of Monomers and Polymers • Polymers tend to have totally different physical and chemical properties from their monomers.
Videos • The Polymer Party – • http://www.youtube.com/watch?v=SgWgLioazSo