Midterm Policies and Exam Topics Overview
This guide covers exam policies, office hour changes, weak acids, amino acids, peptides, sequencing, purification, protein structure, regulation, myoglobin/hemoglobin, carbohydrates, and lipids. Learn about critical concepts for your midterm preparation.

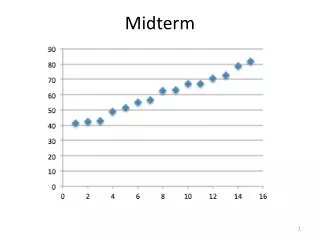
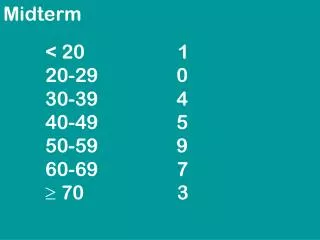
Midterm Policies and Exam Topics Overview
E N D
Presentation Transcript
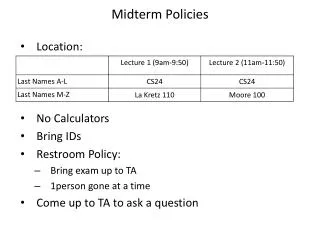
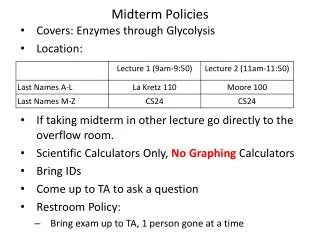
Midterm Policies • Location: • No Calculators • Bring IDs • Restroom Policy: • Bring exam up to TA • 1person gone at a time • Come up to TA to ask a question
Office Hour Changes:in 4077A Young Hall • This week ONLY • Canceled: Fri 10-11am • Additional: Thurs 4-6pm
Weak Acids The weak acid H3PO4has 3 ionizablehydrogens, with the pKas: 2.15, 6.82, 12.38 draw the titration curve for: H+ vs pH pOHvs H+ you make a solution by adding 0.5moles of HPO42- and 0.1 moles of PO43- to 1 Liter of water What is the pH of the solution? Is it a good buffer? You add 0.3 moles of NaOH. What is the new pH of the solution? Is it still a good buffer?
Amino acids/Peptides • pKas: COOH=2, Lys = 10.5, Glu = 4.07, NH3=9.5 • Draw the following peptide at physiological pH: Lakes • What is the net charge of the peptide? • What is the pI of the peptide? • Draw the titration curve for Glutamate (pH vs OH)
Sequencing:What are these compounds and what do they do? LiBH4
Sequencing • Chymotrypsin: • 1: Ile-Ser • 2: Leu-Lys-Val-Phe • 3: Ala-Arg-Gly-Try • Trypsin: • 1: Gly-Trp-Leu-Lys • 2: Val-Phe-Ile-Ser • 3: Ala-Arg
Purification • What are the 5 characteristics that we can use to separate proteins from each other? • What process can be used for each characteristic?
Protein structure • What are the levels of protein structure? • For each: • What interactions stabilize them? • How can we determine the structure? • Diagram Alpha helices and beta sheets • How do you get amphipathic structures in each?
Protein regulation • What is cooperativity? • What is allosteric regulation? • What are the regulators of Hb? And what type of regulators are they?
Myoglobin/Hemoglobin • How do Mb and Hb differ in how they bind oxygen? • How does this relate to their different biological functions? • How does destabilizing the T-state affect oxygen binding to Hb? • How does destabilizing the R-state affect oxygen binding to Hb?
Carbohydrates • What is the structure of glycogen? • What is its biological function and how does its structure relate to this? • How is the structure of Cellulose different from Glycogen? • How is this better for its biological function?
Lipids • How are triacylglycerols better suited for long term energy storage than glycogen? • A fish has traveled to colder water. How would it change the composition of its membranes?