Weather Vs. Climate
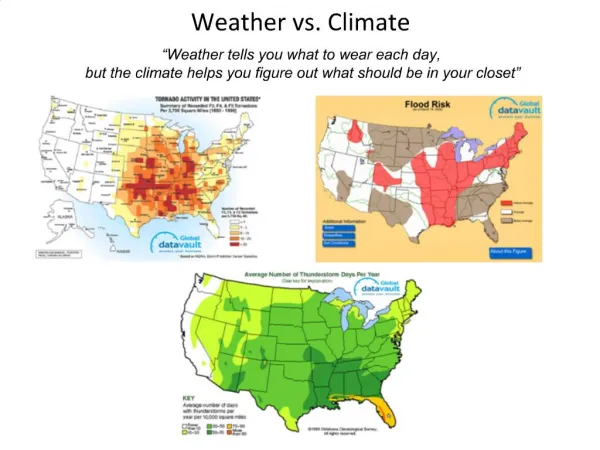
Weather Vs. Climate. Weather is the set of environmental conditions that occur day to day or even minute to minute Climate is the set of environmental conditions that is expected to occur for a given area over any given year. The Earth’s Energy Balance.

Weather Vs. Climate
E N D
Presentation Transcript
Weather Vs. Climate • Weather is the set of environmental conditions that occur day to day or even minute to minute • Climate is the set of environmental conditions that is expected to occur for a given area over any given year
The Earth’s Energy Balance • In order for Earth to maintain its overall climate it must stay within a relatively small temperature range • If earth loses more energy than energy that it gains it will cool • If earth gains more energy than it loses it will heat up • The mechanism that allows earth to remain in this equilibrium is called an energy balance • Because earth’s temperature remains reasonably consistent we can deduce that the energy coming into earth must equal the energy leaving earth
Transfer of Energy • There are three ways energy can be transferred from one object to another Radiation: • The transfer of energy in which atoms or molecules emit electromagnetic waves • These waves carry the energy through space and deposit it only when they interact with some form of matter • Radiant Energy from the Sun (short wave) travels long distances until it interacts with solid matter on Earth. Earth then radiates energy back into space (long wave) as it is heated by the sun
Transfer of Energy Convection/Advection • The transfer of energy by the movement of particles in a fluid (liquid or gas) • Convection is vertical transfer advection is horizontal transfer • Hot air rises This happens because if air is warmer than its surroundings it becomes less dense. As it becomes less dense it will “float” on the more dens air around it – Convection!
Transfer of Energy Conduction: • The transfer of energy in which highly energetic atoms or molecules from one object collide with less energetic atoms or molecules from another object giving them some of their energy • In this case the atoms will stay in their original locations but the energy is allowed to move by this contact
Reflection and Absorption of Energy Reflection • As the Sun’s rays reach earth some of the energy is reflected by the ocean, atmosphere, clouds, snow, ice • The amount of reflectivity of a surface called it’s Albedo Absorption An object or material that absorbs energy and becomes warmer is called a heat sink The heat capacity of an object is the measure of how much heat a substance requires to raise its temperature or how much energy it releases as it cools Heat capacity is measured in joules of energy per kilogram degree J/(Kg ºC).
Albedo Calculations • The amount of energy absorbed by any surface is calculated by: Amount of energy (J) x (1-albedo) • For example if a glacier is receiving 10 000 J of energy over a square meter and the albedo is 0.4, the actual amount of energy absorbed is: 10 000 J x (1-0.4) = 6 000 J Now calculate the amount of energy absorbed by a parking lot with an albedo of 0.02 and receiving 8000 J/m2
Specific Heat Equation • The amount of heat energy (q) gained or lost by a substance is equal to the mass of the substance (m) multiplied by its specific heat capacity (C) multiplied by the change in temperature (final temperature - initial temperature = t) Q = m c t
Specific Heat Questions REMEMBER GRASP PROBLEM SOLVING! 1. How much energy is required to bring 1kg (1L) of water from 20 C to a boil? Note the Specific Heat Capacity of water is 4186 J/Kg C 2. How hot will a 50Kg rock in the dessert get if it receives 592.5 KJ of energy from the sun (C= 790J/Kg C)
Using Heat Capacity and Albedo • Now that we understand Heat Capacity and can calculate the amount of energy absorbed by a surface we can use the equation for heat capacity to predict the temperature change of a surface Assuming the asphalt in the a previous question had a mass of 84kg, how much would you expect the surface temperature to rise?
Putting it all together The passive solar panels connected to a backyard swimming pool have and Albedo of 0.05. The panels will receive 40000 KJ of energy from the sun per hour on an average sunny day. Assuming no heat loss from the pool water to the atmosphere how much might the temperature of the pool be raised in a typical afternoon (4 hours). The volume of an average sized swimming pool is 36 m3 (36000 Kg).