The Global Methane Cycle
The Global Methane Cycle. Methane. CH 4 Colorless gas Component of the atmosphere Main component of natural gas CH 4 + 2O 2 → CO 2 + 2H 2 0 . Why is methane important?. Clean fuel source It is a powerful greenhouse gas It removes Hydroxyls from the troposphere

The Global Methane Cycle
E N D
Presentation Transcript
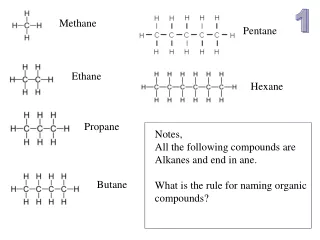
Methane • CH4 • Colorless gas • Component of the atmosphere • Main component of natural gas • CH4 + 2O2→CO2 + 2H20
Why is methane important? • Clean fuel source • It is a powerful greenhouse gas • It removes Hydroxyls from the troposphere • Affects concentrations of water vapor and ozone in the stratosphere • Used in the industrial production of hydrogen, methanol, acetic acid, and acetic anhydride
Methane as a fuel source • Used as a fuel source for electricity generation, heating, and cooking • Methanol is used in vehicles • Heat of combustion is about 802 kJ/mol • Highest heat to weight ratio • Produces the least CO2 per unit of heat
How much methane is there? • ~350 ppb 18,000 years ago • Today atmospheric levels are ~1800 ppb and rising • The most abundant greenhouse gas after CO2 and H2O vapor • Most abundant trace gas in the atmosphere
Where is methane from? • It is produced by organisms and geological processes • Livestock produces 37% of all human-induced methane • Most is released from wetlands in the northern hemisphere and tropics
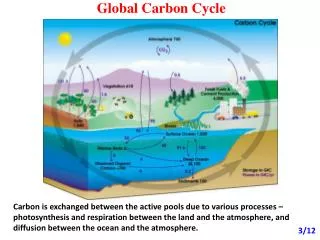
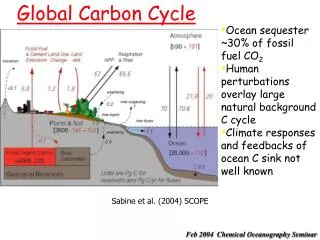
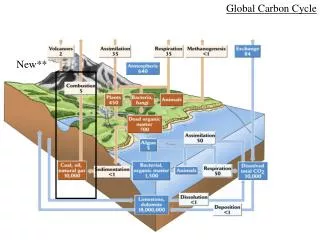
Methane is cycled • Certain organisms and geological processes produce methane • Other organisms and processes use methane and release new products • Several organisms and processes are involved • The methane cycle is part of the larger carbon cycle
Sources: Natural Wetlands Termites Oceans Hydrates & Clathrates
Wetlands • Produces 225 Tg per year. • 50% of wetlands are peat rich. • Produced by methanogenic bacteria decomposing organic materials in anaerobic environments. • CH4 produced in sediments are diffused to the surface via the water column, gas bubbles, or plants. • C6H12O6 -> 3CO2 + 3CH4 • Massive amounts of CH4 trapped in permafrost could be released if temperatures rise.
Termites • Produces 20 Tg per year • Concentrated mostly in grasslands or forests • Produced in the breakdown of cellulose by CH4 oxidizing bacteria
Oceans • Produces 15 Tg per year • Sources are poorly known. • Coastal areas have higher but more variable concentrations. • Produced by seepage areas in seabed with organic rich nutrients.
Hydrates • Produces 10 Tg per year • CH4 Hydrates are ridgid water cages surrounding CH4 molecules. • Mostly on the continental shelf of all latitudes. • Hydrate stability requires high pressures and cold temperatures. Most hydrates occur at depths and regions insulated from climate change. • Climate warming would lead to destabilization of hydrates and release CH4 molecules.
Sources: Anthropogenic • Rice Cultivation • Fossil Fuels • Biomass Burning • Landfills
Rice Cultivation • Produces 100 Tg per year • Production has increased over 40% since the 1980’s. • Produced by anaerobic consumption of organic material by methanogenic bacteria
Domestic Animals • Produces 115 Tg per year • 75% are produced from cows • Produced from fermentation of carbohydrates in the rumen. Microbes in the rumen are capable of breaking down cellulose. • Quanity & Quality of feed, plus weight, age, activity level, and species affect how much CH4 is produced.
Fossil Fuels • Produces 110 Tg per year • Coal Gas and Natural Gas consist almost entirely of methane. • Fossil Fuel sources are coal mining as well as exploration, production, transmissions.
Biomass Burning • Produces 40 Tg per year • CH4 is released when vegetation is burned. • Amount produced depends on the burning technology, temperature, moisture and carbon content in the vegetation.
Landfills • Produces 40 Tg per year • Decomposition of biodegradable organic material in landfills produces CO2 and CH4.
Sinks Losses of CH4 occur in lithosphere and the atmosphere Lithosphere: Dry soil oxidation Atmosphere: Trophospheric reactions with OH and losses to the stratosphere
Dry Soil Oxidation • A loss of 30 Tg per year • Can act as an sink for both atmospheric CH4 and CH4 produced in deeper soil layers. • Methanotrophs use CH4 as a source of carbon in a process called methane oxidation • Methanotrophs exist in two forms: 'high capacity - low affinity' methanotrophs ’low capacity - high affinity' methanotrophs Changes in land use practices, better fertilizer application, and land conversion may help prevent the loss of methane sinks.
Tropospheric Reactions With OH • A loss of 510 Tg per year • The most dominant form of CH4 loss. • OH + CH4 -> CH3 + H2O • CH4 is removed when it reacts with the hydroxyl radical OH. This happens when cosmic rays strike a water vapor molecule. • Hydroxyl Radicals are considered the “Detergent” of the atmosphere because they react with many pollutants.
Losses To The Stratosphere • A loss of 40 Tg per year • Plays a minor role in removing CH4 from the atmosphere • CH4 is loss to the stratosphere when it reacts with OH, Cl and O(1D) • The stratosphere is very dry and any water vapor produced from methane oxidation becomes a greenhouse gas itself.
General Facts about Methane • Known as a ‘well-mixed greenhouse gas’ because of long lifetime so it is able to evenly distribute in atmosphere • More potent than CO2, but concentration of CO2 is higher so CO2 has greater effect on global warming -Control of methane emissions turns out to be a more powerful lever to control global warming than anticipated
Effects on the Environment • Methane is a greenhouse gas ->absorbs heat in the atmosphere->Increase in temperature • Since Industrial Revolution, atmospheric methane concentration has doubled • Contributes about 20% towards the greenhouse effect, second to CO2
Controversy • Levels of Greenhouse gases can be determined by: -measuring level of gases after mixing with other gases in the atmosphere (official IPCC) OR -measuring level of gases before entering atmosphere.
Prevention for the Future • Methane to Market (United States, the UK, China, Russia, Brazil, India, Italy, Japan, Australia and Nigeria) Goal: To recover Methane and use it as clean energy.
Federal Clean Air Act Collection of methane gas from landfills and treated • Processing manure using anaerobic digesters making methane available for conversion to useful energy • New rice harvesting method, reduce methane gas, increase yield
Methanogenesis - The Big Picture Why is this process interesting? • Terminus of anaerobic food chain. • Prevents sequestering of large amts or organic material in anaerobic ecosystems. • Potential natural gas source. • Methanogens are the most important CO2- reducing prokaryotes, but are also a source of greenhouse gas (CH4).
Methanogenesis within the Carbon Cycle. Water column sediments
Methanogenesis: The Process Overall Reaction: 4 H2 + H+ + HCO3- CH4 + 3H2O
And who are these? • METHANOGENS • Archaea, ubiquitous in highly reducing habitats, also found in some “oxic” habitats • Obligate anaerobes • H2 + CO2 --> methane (CH4) • Autotrophic (fix CO2), chemolithotrophic (use inorganic H2 as e- donor) • Some use endproducts of fermentation (acetate, etc.) • Some important genera: Methanococcus, Methanosarcina • Capable of using elemental Sulfur as a terminal e- acceptor (anaerobic respiration - sulfur reduction) • METHANOTROPHS • Bacteria • Aerobes • Utilize methane: • CH4 + O2 --> CO2 + H2O • Rxn happens at interface btwn anoxic and oxic layers of soil (as CH4 diffuses up). • Genus: Methylomonas
Cool and current studies A new biogeochemical cycling pathway couples anaerobic methane oxidation to denitrification. (Nature; vol. 440, 04/06). • Freshwater sediments receive high loads of anthropogenic NO3- and CH4. • Theoretically microbes should be able to use NO3-/NO2- to anaerobically oxidize CH4, but prior to this report it has not been shown. • This study showed direct, anaerobic oxidation of methane coupled to denitrification in the complete absence of O2. CH4 + NO3-/NO2- + H+ ---> CO2 + N2 + H2O • consortium consisted of two microbes: an uncultured bacteria and an archaea related to marine methanotrophic Archaea. Relevance: • new pathways fill in holes in our understanding of global biogeochemical cycling.
Methanogens on Mars? FEMS Micro. Ecol. (2007) Greenland Ice Sheet • 10 ppbv CH4 on mars, estimated 300 tons lost/year - 270 tons/year must be generated to offset loss. • Is this biogenic or abiogenic methane? • Metabolic rates of methanogens in extreme cold on Earth can be used to estimate metabolic rates and cell densities of martian methanogens.
Conclusions • Methane is the simplest hydrocarbon • Methane is a clean fuel source • Methane is a powerful greenhouse gas • Methane is part of the carbon cycle
Conclusions • The major reservoirs of CH4 are fossil fuel reservoirs, hydrates and Clathrates, and the atmosphere • Wetlands are the dominant natural source of CH4 • Domestic animals are the largest anthropogenic source of CH4 • The Hydroxyl Radical OH, is the dominant form of CH4 loss. • There has been an increase in terrestrial sources and a decrease in sinks, leading to excess CH4 in the atmosphere
Conclusions • Reducing Methane will have a stronger influence in reduce Greenhouse effect • ‘Methane To Market’ and ‘Federal Clean Air Act’ hopes to reduce Methane output • New agriculture and use of anaerobic digesters also reduce Methane output
Conclusions • The methane cycle is composed of two groups: methanogens (produce methane) and methanotrophs (consume methane). • Methanogenesis is carried out anaerobically by Archaea, methanotrophy is carried out aerobically by bacteria. • Methanogens are ubiquitous in anoxic environments (wetlands, sediments, etc.) as well as some oxic ones.
REFERENCES • Madigan, Martinko, and Parker. 1997. Brock Biology of Microorganisms. 8th ed. Prentice-Hall, New Jersey. • Stanier, R.Y., Ingraham, J.L., Wheelis, M.L., and Painter, P.R. 1986. The Microbial World. 5th ed. Prentice-Hall, New Jersey. • Strous, M. et al. 2006. A microbial consortium couples anaerobic methane oxidation to denitrification. Nature, 440:918-921. • Price, P.B. 2007. Microbial life in glacial ice and implications for a cold origin of life. FEMS Microbiol. Ecol. 59:217-231. • http://www.newscientist.com/article.ns?id=mg18725124.500 • http://www.meteor.iastate.edu/gcp/studentpapers/1996/atmoschem/brockberg.html • http://www.epa.gov/methane/sources.html • http://www.ghgonline.org/methanesinksoil.htm • http://www.igac.noaa.gov/newsletter/21/methane_sink.php • http://earthobservatory.nasa.gov/Newsroom/NewImages/images.php3?img_id=16827 • http://www.grida.no/climate/ipcc_tar/wg1/134.htm#4211
References • http://www.scienceagogo.com/news/20050625005443data_trunc_sys.shtml • http://www.ace.mmu.ac.uk/eae/Global_Warming/Older/Methane.html • http://egov.oregon.gov/ENERGY/RENEW/Biomass/Environment.shtml • http://www.ens-newswire.com/ens/jul2005/2005-07-19-01.asp