ORGANIC CHEMISTRY
1.23k likes | 3.19k Views
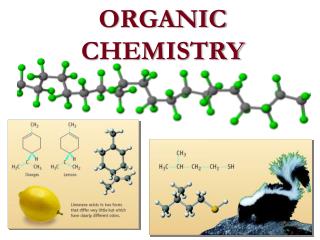
ORGANIC CHEMISTRY. Naming Saturated Hydrocarbons. The International Union of Pure and Applied Chemistry (IUPAC) names for the first 12 "straight-chain" or "normal" alkanes are:. Alkanes and Cycloalkanes. The simplest saturated hydrocarbons are called alkanes.

ORGANIC CHEMISTRY
E N D
Presentation Transcript
Naming Saturated Hydrocarbons • The International Union of Pure and Applied Chemistry (IUPAC) names for the first 12 "straight-chain" or "normal" alkanes are:
Alkanes and Cycloalkanes • The simplest saturated hydrocarbons are called alkanes. • Methane, CH4, is the simplest alkane. • The alkanes form a homologous series. • Each member of the series differs by a specific number and kind of atoms.
Alkanes and Cycloalkanes • The alkanes differ from each other by a CH2 or methylene group. • All alkanes have this general formula. CnH2n+2 • For example ethane, C2H6 , and propane, C3H8 , are the next two family members.
Alkanes and Cycloalkanes • Isomers are chemical compounds that have the same molecular formulas but different structures. • Two alkanes have the molecular formula C4H10. • They are a specific type of isomer called structural isomers. • Branched-chain alkanes are named by the following rules.
Naming Saturated Hydrocarbons • Choose the longest continuous chain of carbon atoms which gives the basic name or stem. • Number each carbon atom in the basic chain, starting at the end that gives the lowest number to the first group attached to the main chain (substituent). • For each substituent on the chain, we indicate the position in the chain (by an Arabic numeric prefix) and the kind of substituent (by its name). • The position of a substituent on the chain is indicated by the lowest number possible. The number precedes the name of the substituent. • When there are two or more substituents of a given kind, use prefixes to indicate the number of substituents. • di = 2, tri = 3, tetra = 4, penta = 5, hexa = 6, hepta = 7, octa = 8, etc. • The combined substituent numbers and names serve as a prefix for the basic hydrocarbon name. • Separate numbers from numbers by commas and numbers from words by hyphens. • Words are "run together".
Naming Saturated Hydrocarbons • Alkyl groups (represented by the symbol R) are common substituents. • Alkyl groups are fragments of alkanes in which one H atom has been removed for the connection to the main chain. • Alkyl groups have the general formula CnH2n+1. • In alkyl groups the -ane suffix in the name of the parent alkane is replaced by -yl. • A one carbon group is named methyl. • A two carbon group is named ethyl. • A three carbon group is named propyl. • Three alkanes have the formula C5H12. • There are three structural isomers of pentane. • n-pentane 2-methylbutane 2,2-dimethylpropane
Alkanes and Cycloalkanes • There are five isomeric hexanes, C6H14. n-hexane 2-methylpentane 3-methylpentane 2,2-dimethylbutane 2,3-dimethylbutane • The number of structural isomers increases rapidly with • increasing numbers of carbon atoms. • The boiling points of the alkanes increase with molecular weight.
Alkanes and Cycloalkanes • Cyclic saturated hydrocarbons are called cycloalkanes. • They have the general formula CnH2n. • Some examples are: cyclopentane cyclooctane
Alkenes • The three classes of unsaturated hydrocarbons are: • alkenes and cycloalkenes, CnH2n • alkynes and cycloalkynes, CnH2n-2 • aromatic hydrocarbons • The simplest alkenes contain one C=C bond per molecule. • The general formula for simple alkenes is CnH2n. • The first two alkenes are: • Each doubly bonded C atom is sp2 hybridized. • The sp2 hybrid consists of: • two s bonds (single bonds) and • one s and one p bond (double bond)
Alkenes • The systematic naming system for alkenes uses the same stems as alkanes. • In the IUPAC system, the -ane suffix for alkanes is changed to -ene. • Common names for the alkenes have the same stem but use the suffix -ylene is used. • In chains of four or more C atoms, a numerical prefix shows the position of the lowest-numbered doubly bonded C atom. • Always choose the longest chain that contains the C=C bond. • Polyenes contain two or more double bonds per molecule. • Indicate the number of double bonds with suffixes: • -adiene for two double bonds. • -atriene for three double bonds, etc. • The positions of the substituents are indicated as for alkanes. • The position of the C=C bond(s) is/are given the lowest number(s) possible. 1,3-hexadiene 1,2,5-hexatriene 2,3-dimethyl-1,3,5-hexatriene
Cycloalkenes • Cycloalkenes have the general formula CnH2n-2. • Examples are: • cyclopentene • cyclohexene
Alkynes • Alkynes contain CC bonds. • The simplest alkyne is C2H2, ethyne, or acetylene. • Alkynes with only one C C bond have the formula CnH2n-2. • Each carbon atom in a C C bond is sp hybridized. • Each sp hybrid contains two bonds and two bonds. • The carbon atom will have one single bond and one triple bond. • Alkynes are named like the alkenes except that the suffix -yne is used with the characteristic stem • The alkyne stem is derived from the name of the alkane with the same number of carbon atoms. 3-heptyne 2-octyne
Aromatic Hydrocarbons • Historically, aromatic was used to describe pleasant smelling substances. • Now it refers to benzene, C6H6,and derivatives of benzene. • Other compounds that have similar chemical properties to benzene are also called aromatic. • The structure of benzene, C6H6, is: • Coal tar is the common source of benzene and many other aromatic compounds. • Some aromatic hydrocarbons that • contain fused rings are: • napthalene
Resonance in Benzene • C6H6 has two resonance structures with alternating double bonds. • The π electrons are delocalized over the ring. • C–C single bond = 154 pm • C=C bond = 134 pm • CC bonds in benzene = 139 pm Resonance structures of benzene, C6H6 Abbreviated representation of resonance structure π electrons delocalized
Other Aromatic Hydrocarbons • Many aromatic hydrocarbons contain alkyl groups attached to benzene rings (as well as to other aromatic rings). • The positions of the substituents on benzene rings are indicated by the prefixes: • ortho- (o-) for substituents on adjacent C atoms • meta- (m-) for substituents on C atoms 1 and 3 • para- (p-) for substituents on C atoms 1 and 4 m-xylene o-xylene p-xylene
General Properties and Reactivity of Alkanes, Alkenes, and Alkynes • Alkanes Consist of C–C and C–H bonds that are strong, not polar, and not easily attacked by nucleophiles or electrophiles, so reactivity is limited • The multiple bond of an alkene produces geometric isomers (cis and trans) • Cis and trans isomers of alkenes behave as distinct compounds with different chemical and physical properties • The hydrogen atom of a terminal alkyne can be removed as H+,forming anacetylide ion (R–CC–) • Acetylide ions are potent nucleophiles used for making longer carbon chains by a nucleophilic substitution reaction • Rotation about the carbon-carbon multiple bonds of alkenes and alkynes cannot occur without breaking a bond, which constitutes a large energy barrier to rotation. Alkenes and alkynes are prepared by elimination reactions • Arenes undergo substitution rather than elimination due to stability from delocalization of their electron density, and are poor nucleophiles
Organic Halides • A halogen atom may replace almost any hydrogen atom in a hydrocarbon. • The functional group is the halide (-X) group. • Examples include: • chloroform, CHCl3 • 1,2-dichloroethane, ClCH2CH2Cl • para-dichlorobenzene
Alcohols and Phenols • The functional group in alcohols and phenols is the hydroxyl (-OH) group. • Alcohols and phenols can be considered derivatives of hydrocarbons in which one or more H atoms have been replaced by -OH groups. • Phenols are derivatives of benzene in which one H has been replaced by replaced by -OH group. • The stem for the parent hydrocarbon plus an -ol suffix is the systematic name for an alcohol. • A numeric prefix indicates the position of the -OH group in alcohols with three or more C atoms. • Common names are the name of the appropriate alkyl group plus alcohol.
Alcohols and Phenols • Ethyl alcohol (ethanol), C2H5OH, is the most familiar alcohol. • Phenol, C6H5OH, is the most familiar phenol.
Alcohols and Phenols • Alcohols can be classified into three classes: • Primary (1°) alcohols like ethanol have the -OH group attached to a C atom that has one bond to another C atom. H3 OH • Secondary(2°) alcohols have the –OH group attached to a C atom that has bonds to 2 other C atoms. • For example,2-propanol: H3 H3 OH • Tertiary (3°) alcohols have the –OH group attached to a C atom that is bonded to 3 other C atoms. • For example, 2-methyl-2-propanol H3 H3 OH H3
Alcohols and Phenols • Alcohols are named using the stem for the parent hydrocarbon plus an -ol suffix in the systematic nomenclature. • A numeric prefix indicates the position of the -OH group in alcohols with three or more C atoms. • Common alcohol names are the name of the appropriate alkyl group plus the word alcohol. 1-pentanol 2-pentanol 3-pentanol 1-pentyl alcohol 2-pentyl alcohol 3-pentyl alcohol
Alcohols and Phenols • There are several isomeric monohydric acyclic (contains no rings) alcohols that contain more than three C atoms. • There are four isomeric four-carbon alcohols. 1-butanol 2-butanol 2-methyl-1-propanol 2-methyl-2-propanol
Alcohols and Phenols • There are eight isomeric five-carbon alcohols.
Alcohols and Phenols • Polyhydric alcohols contain more than one -OH group per molecule.
Alcohols and Phenols • Phenols are usually called by their common (trivial) names.
Ethers • Ethers may be thought of as derivatives of water in which both H atoms have been replaced by alkyl or aryl groups. • Ethers are not very polar and not very reactive. • They are excellent solvents. • Common names are used for most ethers.
General Properties and Reactivity of Alcohols and Ethers • Alcohols and Ethers • Have “bent” structures and are able to hydrogen-bond • Are good solvents for organic compounds • Alcohols are prepared by • the addition of water to the carbons of a double bond or by substitution of an alkyl halide by hydroxide, a potent nucleophile • he reduction of compounds containing a carbonyl functional group ( CO) • Undergo two major types of reactions: those involving cleavage of the O–H bond, which produces an acid, and those involving cleavage of the C–O bond occurring under acidic conditions where the –OH is first protonated followed by a nucleophilic substitution • Phenols are more acidic than alcohols because of interactions between the oxygen atom and the ring • Ethers are prepared by • a substitution reaction in which the highly nucleophilicalkoxide ion (RO–) attacks the carbon of the polarized C–X bond of an alkyl halide (R´ X) • Unreactive because they lack the –OH unit
Aldehydes and Ketones • The functional group in aldehydes and ketones is the carbonyl group.
Aldehydes and Ketones • Except for formaldehyde, aldehydes have one H atom and one organic group bonded to a carbonyl group. • Ketones have two organic groups bonded to a carbonyl group.
Aldehydes and Ketones • Common names for aldehydes are derived from the name of the acid with the same number of C atoms. • IUPAC names are derived from the parent hydrocarbon name by replacing -e with -al.
Aldehydes and Ketones • The IUPAC name for a ketone is the characteristic stem for the parent hydrocarbon plus the suffix -one. • A numeric prefix indicates the position of the carbonyl group in a chain or on a ring.
General Properties and Reactivity of Aldehydes and Ketones • Aldehydes and ketones • Contain the carbonyl functional group • Are prepared by • the oxidation of alcohols • reducing a carboxyl group (–CO2H) to a carbonyl group, which requires a good reducing agent • Characterized by nucleophilic attack at the carbon atom of the carbonyl functional group and electrophilic attack at the oxygen atom • React with organometallic compounds that contain stabilized carbanions such as the Grignard reagents (RMgX, where X = Cl, Br, ), which convert the carbonyl functional group to an alcohol and lengthen the carbon chain • Aromatic aldehydes have intense and characteristic flavors and aromas, and many ketones also have intense aromas • Ketones are found in hormones responsible for sex differentiation in humans
Amines • Amines are derivatives of ammonia in which one or more H atoms have been replaced by organic groups (aliphatic or aromatic or a mixture of both). • There are three classes of amines.
General Properties and Reactivity of Amines • Tertiary amines form cations in which all four H atoms are replaced by alkyl groups and are called quaternary ammonium salts, which can be chiral if all four substituents are different • Alkylamines can be prepared by nucleophilic substitution reactions of polar alkyl halides with ammonia or other amines • Reactions of amines are dominated by two properties: • the ability of amines to act as weak bases and • their tendency to act as nucleophiles, both resulting from the lone pair of electrons on the nitrogen atom • Amines behave as bases by accepting a proton from an acid to form an ammonium salt • Amines can react with any electrophile • Aryl amines are weaker bases than alkylamines because the lone pair of electrons on nitrogen interacts with the bonds of the aromatic ring
Carboxylic acids contain the carboxyl functional group. The general formula for carboxylic acids is: R represents an alkyl or an aryl group Carboxylic Acids • IUPAC names for a carboxylic acid are derived from the name of the parent hydrocarbon. • The final -e is dropped from the name of the parent hydrocarbon • The suffix -oic is added followed by the word acid. • Many organic acids are called by their common (trivial) names which are derived from Greek or Latin.
Carboxylic Acids • Positions of substituents on carboxylic acid chains are indicated by numeric prefixes as in other compounds • Begin the counting scheme from the carboxyl group carbon atom. • They are also often indicated by lower case Greek letters. • = 1st C atom • = 2nd C atom • = 3rd C atom, etc.
Nomenclature of Carboxylic Acids • Dicarboxylic acids contain two carboxyl groups per molecule.
Carboxylic Acids • Aromatic acids are usually called by their common names. • Sometimes, they are named as derivatives of benzoic acid which is considered to be the "parent" aromatic acid.
General Properties and Reactivity of Carboxylic Acids • Can be prepared from the oxidation of alcohols and aldehydes or through the reaction of a Grignard reagent with CO2, followed by acidification • Reactions of carboxylic acids are dominated by their polar carboxyl group and their acidity • Reactions with strong bases produce carboxylate salts • Less susceptible to nucleophilic attack due to delocalization of bonding over three atoms (O–C–O) • Substitution of the –OH of a carboxylic acid produces derivative compounds with different tendencies to participate in resonance with the CO functional group • Resonance structures have significant effects on the reactivity of carboxylic acid derivatives, but their influence varies, being least important for halides and most important for the nitrogen of amides • Two important carboxylic acid derivatives are esters and amides
General Properties and Reactivity of Ester Carboxylic Acid Derivatives • Have the general formula RCO2R´, where R and R´ can be any alkyl or aryl group • Prepared by the reaction of an alcohol (R´OH) with a carboxylic acid (RCO2H) in the presence of a catalytic amount of strong acid (an electrophile); this protonates the doubly bonded oxygen atom of the carboxylic acid (a nucleophile) to give a species that is more electrophilic than the parent carboxylic acid • The nucleophilic oxygen atom of the alcohol attacks the electrophilic carbon atom of the carboxylic acid and a new C–O bond is formed –General overall reaction OH+ O R–C + R´OH R–C + H2O OH OR´ –If an ester is heated with water in the presence of a strong acid or base, the reverse reaction will occur, producing the parent alcohol, R´OH, and either the carboxylic acid, RCO2H (under acidic conditions), or the carboxylate anion, RCO2– (under basic conditions)
General Properties and Reactivity of Amide Carboxylic Acid Derivatives – The two substituents on the amide nitrogen can be hydrogen atoms, alkyl groups, aryl groups, or any combination of two of those species O R1–C–N–R2 R3 – Are prepared by the nucleophilic reaction of amines with other, more electrophilic carboxylic acid derivatives, such as esters – Are unreactive because of bonding interactions between the lone pair of electrons on nitrogen and the carbonyl group, which inhibits free rotation about the C–N bond – Stability of amide bond is important in biology because they form the backbones of peptides and proteins
When compounds contain more than one functional group, the order of precedence determines which groups are named with prefix or suffix forms. The highest precedence group takes the suffix, with all others taking the prefix form. However, double and triple bonds only take suffix form (-en and -yn) and are used with other suffixes.
Reactivity of Organic Molecules • The reactivity of a molecule is affected by the degree of substitution of the carbon bonded to a functional group; the carbon is designated as primary, secondary, or tertiary – Primary carbon is bonded to only one other carbon and a functional group – A secondary carbon is bonded to two other carbons and a functional group – A tertiary carbon is bonded to three other carbons and a functional group • Identifying the transient species formed in a chemical reaction, some of which are charged, enables chemists to predict the mechanism and products of the reaction
Reactive Intermediates • When cleaving a C–H bond, the most common species formed is C+,called a carbocation, which has only six valence electrons and is electron deficient – A carbocation is an electrophile, a species that needs electrons to complete its octet – A tertiary carbocation is more stable than one that is primary because it increases electron density at the carbocation • Adding an electron to a free radical produces a carbanion, a negatively charged carbon with eight valence electrons • A carbanion is a nucleophile, an electron-rich species • Carbanions are destabilized by groups that donate electrons, so a tertiary carbanion is less stable than a primary one
Common Organic Reactions • Five common types of organic reactions: • Substitution (SN1, SN2)– one atom or group of atoms in a substance is replaced by another atom or group of atoms from another substance. A typical substitution reaction is the reaction of hydroxide ion with methyl chloride (nucleophilic substitutionreactions): CH3Cl + OH– CH3OH + Cl • Elimination (E1, E2)– in which adjacent atoms are removed, or “eliminated,” from a molecule with the formation of a multiple bond and a small molecule are called elimination reactions • Addition – the components of a species A–B are added to adjacent atoms across a carbon-carbon multiple bond is called an addition reaction HCl + CH2CH2 CH3CH2Cl • Free-radical reactions – the best known is the reaction of a saturated hydrocarbon with a halogen: CH3CH3 + Br2 CH3CH2Br + HBr • Free radical reactions occur in three stages: initiation,propagation, and termination • At high temperature or in the presence of light, the weak Br–Br bond generates Br atoms • A bromine atom attacks ethane, producing a free radical, which reacts with a bromine molecule • combination of two bromine atoms, of two ethyl radicals, or of an ethyl and a bromine radical • Oxidation-reduction reactions – are common in organic chemistry and can be identified by: • An increase in either is an oxidation, whereas a decrease is a reduction • An increase in the number of hydrogens in a hydrocarbon is an indication of a reduction • In compounds with a carbon-nitrogen bond, the number of bonds between the C and N atoms increases as the oxidation state of the carbon increases A B CH2–CH2 CH2CH2 + A–B