Sponsored Programs Administration Pre- Award Training
Sponsored Programs Administration Pre- Award Training. October, 2005. Why are we here?. Learn more about what goes on the grant admin world Get some tips and tools on how better to do our jobs Understand who I need to work with at SPA, when and how . Specific Tasks.

Sponsored Programs Administration Pre- Award Training
E N D
Presentation Transcript
Sponsored Programs Administration Pre- Award Training October, 2005
Why are we here? • Learn more about what goes on the grant admin world • Get some tips and tools on how better to do our jobs • Understand who I need to work with at SPA, when and how
Specific Tasks • How do we manage the proposal process? • How do I find a sponsor? • How should we construct our budgets? • What do I need to do once we receive an award?
The SPA Office • Organizational chart • Realignment of resources • Director for Pre-Award Services – Carole Bach • Director of Research Admin – Marlene Erno (Institutional Support) and Gail Ryan (Operations)
Other Presenters • Vance Briceland, Information Officer • Gail Ryan, Director of Post-Award Services and friends • Post Award Presenters
What is SPA’s mission? • Ensure consistency, reasonableness, allocability and compliance with institutional and agency policies • Protect integrity of institution in its management of sponsored programs • Provide the fiduciary review and management (Stewardship) of sponsor’s funds
Definitions • Reasonableness • Nature of goods or services acquired reflect the action that a prudent person would have taken under the same exact circumstances • The expense is necessary for the grant’s performance, the recipient followed organizational policies • Acted with due prudence in carrying out their responsibilities
Definitions • Allocability • Cost incurred solely for purposes of the grant • Expense is clearly identified with the project • Conformance • Reasonable and Necessary • In conformance with limitations/exclusions • Consistency • Treated in the same manner across all proposals and awards
What is College/Dept’s role? • Proper Budgeting Development and Management • Adhere and ensure compliance with unit’s policies • Provide day-to-day management of sponsor funds • Provide first line of support and communication to faculty and program staff
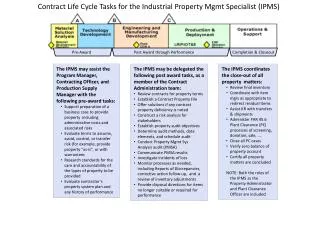
The Grant Life Cycle • From when faculty has idea until the final report has been submitted … and, then some • Extremely variable in timeframe, process, requirements • Need to pay attention at all stages
Where do ideas come from? • The “When I was a child …” model • The “I had a family member …” model • The “I had a college professor …” model • Why is this important? – The passion of researcher
Find the money! • There are many sources for money • Identifying the right match(es) for their work • Role of Vance Briceland – Information Officer
Sponsor Missions • Governmental Sponsors exist to fulfill legislated or mandated goals • Foundation fund projects which support initiative identified in their charters • Not-for Profits support special purposes, e.g American Cancer Society, etc
When do we submit? • RFP’s, RFA’s and Program announcements • Regular scheduled cycles – NIH, foundations, organizations • As part of larger proposal – “we, the subcontractor” • Proposals with no deadlines • Pre-Proposals
How long does it take to build a proposal? • Should the question be “how long should it take?” • Break proposal into manageable components • Manage each piece individually • Allow time for adequate reviews (internal and external) • The answer - ????
Am I in the game? • Acknowledgement from sponsor • Assignment to study section (NIH) • Peer review by sponsor • 3 6 9 months • Did I really get an answer? • Positive feedback vs. award • The tentative process
Receiving Award • Needs to be handled by SPA Pre-Award • SPA Starts wheels turning • Need to begin communication with college/departments • Need to make sure there is a proposal and it matches the award
Award activation • The award has been accepted by the PI • The award has been accepted by WSU • SPA started the process: • Do we have all the “right” pieces • Does it need a new index? • Established on BANNER • SPA has executed a subcontract
Award Life Cycle for SPA • Monitor the award from budgetary perspective • Pay subcontractor, monitor invoices • Manage revenue: • Create invoices • Make LOC draws • Account receivable follow-up • Track other award conditions – reporting, etc.
More Award Life Cycle for SPA • Financial Close-out: • Final invoice or FSR • Indirect Cost reconciliation • Other close out activities: • Intellectual property • Fixed Asset/Property
Award Life Cycle for Dept • Complete the project • Submit non-financial progress reports • Monitor budget activities • Timely Personnel charges • Setting up blank orders or PO’s • Appropriateness of charges
The Standard Proposal Components • Agency Info • Administrative Info • Personnel and Subcontractors • Facilities and Resources • Budget • Protocols (Human Subjects, Animals) • The Science and Abstract
Agency Information • Who is the sponsor of the work? • Who is the prime sponsor of the work? Is this a subcontract? • Is the proposal in response to an RFP or program announcement? • Who is the Principal Investigator and what role do the play
Administrative Components • What is title of program? And, in how many characters may I use to create the title? • Where does it need to be sent? • When is this proposal due? • How many copies and in what format? • How much $$ can we ask for? • What is allowed what isn’t? • What type of work is being proposed: • Research • Instruction • Other Sponsored Activity • Student Service • Clinical Trials • Fellowship
The Standard Proposal Components • Agency Info • Administrative Info • Personnel and Subcontractors • Facilities and Resources • Budget • Protocols (Human Subjects, Animals) • The Science and Abstract
Personnel, Consultants & Subcontractors • Need to identify all WSU employees that will be involved on project • Can use named and unnamed individuals • Identify role for each individual – PI, Research Asst. etc • Identify level of effort for each individual • Any intent to cost share any effort? Is there a requirement?
Am I a consultant or employee? • No WSU employee should be noted as a consultant or subcontractor • IRS has specific rules as to who is consultant and who is employee • Do not confuse with the legal mechanism by which we pay to drive role definition • If person controls work arrangement, they are a consultant
Are they a consultant or a subcontractor? • Are they providing a service beyond expertise, creation of item to be analyzed? • Are they supplying their own facilities and resources? • Are they part of an organization that does task as part of their normal business? • If these are true = subcontractor
What do we need from subcontractor? • A proposal – nice but we won’t get one • Statement of work to be included/merged into science • Proposed budget for work they are proposing • Statement of intent to subcontract
Effort Allocations for Personnel • Identify each person’s expected level of effort for project • Identify the time period for level of effort, be specific to their appointment type • For academic appt, break into AY (9 months) and Summer (3 months) • For fiscal year appointment, use 12 month period
Calculating Effort • Effort the same over 12 months • = % Effort • Effort different for academic year & summer • =(% Effort for academic year x 9 + % effort for summer month x # summer months)/12 • Calculation • 12 month = % Effort * 12 month salary or 9 month salary x 1.33 • Two line entry for 9 month & Summer
The Standard Proposal Components • Agency Info • Administrative Info • Personnel and Subcontractors • Facilities and Resources • Budget • Protocols (Human Subjects, Animals) • The Science and Abstract
Facilities and Resources • Where will the majority of the work take place? Bldg and room • Will there be any need to modify/renovate any of this space? • Will there be any work completed at off-campus facilities? How much? • Will there be any specialized WSU facilities or Service Center used to conduct of work? If so, to what extent would they be used?
Developing the Budget • Step 1 – Review Sponsor/Program Guidelines • What restrictions are invoked? • Any cost sharing requirement? • Ask SPA • Step 2 – Finalize Personnel Component • Is there a salary cap? Current salary cap for NIH is $180,100 • Convert effort to dollars • Apply applicable fringe benefit rates for staff • Add amounts for any consultants
Developing the Budget • Step 3 – Finalize Subcontract Component • Identify direct vs. indirect portions of budget • 8% F & A Costs allowed for International subcontractors on NIH • Add total budget for subcontractors as direct costs in your proposal • Step 4 – Finalize Travel Component • Identify any planned trips • Identify foreign vs. domestic • Estimate cost of each trip, including actual travel, lodging, meals/per diem, registration • Follow WSU Travel Policies
Developing the Budget • Step 5 – Finalize Equipment • What is equipment? $5,000 or more and a useful life of more than 2 years by WSU policy • Does Sponsor have rules? • Step 6 – Supplies • Tangible Property • Supplies for labs, equipment less than $5,000 • Step 7 – Patient Care Costs • Inpatient Costs • Outpatient costs, some of which get billed to 3rd party payers, must be Medicare rate for federal government • Services able to be purchased with purchase requisition are not patient care costs
Developing the Budget • Step 8 – Other Costs • Other services • Unique project needs • Non-Tangible items needed for the project but you can’t see, touch or store it. • Step 9 – Participant Costs • For training programs outside of WSU courses • Travel and subsistence • Considered other costs
Developing the Budget • Step 10 – Training Costs for Training Grants Only • Tuition and Stipends for trainees • Stipends set by grantor • Institutional allowances – used for travel and other allowable costs, i.e. health insurance • Not Employees
Cost Sharing on Grants • Whenever we have a legitimate cost for which the sponsor doesn’t pay, we have cost sharing • Cost sharing is a real cost to the University • Therefore, as good business people, we should minimize cost sharing
Cost Sharing on Grants • Cost sharing may be mandated by sponsor/program guidelines • When cost sharing is mandatory, we should be careful to meet, not exceed, the requirement • Construct a full budget, then determine what should be cost shared
Cost Sharing Types • Mandatory – When agency states in their guidelines that the University must contribute funds to the project • Cash match – University Expenses • 3rd Party – Cost sharing commitment met by someone else outside the University • Voluntary – When the PI includes items in the budget that are not reimbursed by the grant • Salary cap is considered voluntary –These include NIH’s current salary cap as well as some private agencies that have there own salary caps
Budget Justifications • Justifies WHY they need the funding requesting • Should relate back to the work outlined in the technical proposal • Make sure justification matches budget as related to % effort, itemization of costs • At the proposal stage: WSU requires the justification required by the agency
Indirect Costs • Really, they aren’t all that bad • Reimbursement for costs unable to be allocated to individual project • Utilities, building/equipment depreciation • Administrative costs – central and department
Indirect Costs(Facilities and Administrative F & A) • Rates are structured by activity and location • Current rates: on-campus off-campus Research 50.5%* 26%* Instructional 50.5% * 26%* Other 40.0%* 26%* Clinical Trials 26%* *based on Modified Total Direct Costs
What is MTDC ? • There are certain items that are excluded from Facilities & Administrative charges • They are Rent, equipment, tuition, patient care costs, stipends, scholarships and the amount of the subcontract over $25,000 • By subtracting these items from the total direct costs you arrive at Modified Total Direct Costs which is used to calculate F&A amount
Indirect CostsRate Application • Indirect Cost Rates can be set by Program Activity • Normally, Federal pays full load for Research • Normally, State and local government pay reduced rates • Normally, industry should pay full load • Normally, foundations pay reduced rate
Indirect CostsWaivers vs. Reductions • A sponsor may restrict the level of indirect cost we can recover • When sponsor mandated, this is reduction • Sometimes, we “offer/propose” a reduced rate • This is a waiver