Caffeine
E N D
Presentation Transcript
Caffeine Chemistry 11 Block B Organic Molecule Project Kalum Lau
History of Caffeine Before recorded history, caffeine has been used since the Stone Age, humans would chew on certain seeds, barks or leaves knowing it will give them energy. Caffeine was also used for it’s effects in ancient China, when humans realized boiling tea plants or berries to make tea can increase the intensity of alertness. Based on ancient writings, it is said that tea can improve awareness and concentration. However, they didn’t know it was the effects of caffeine. Later during the 1500s the cacao bean known for its effects by caffeine, was used in Mexico by the Olmec's who harvested the plant to make a cold, spicy and unsweetened drink.
History of Caffeine The chemical itself was discovered and purified in 1819 by a physician named Friedlieb Ferdinand Runge. Joahn Wolfgang Van Goethe; a famous poet who admired the chemist Doebereiner and his discoveries, was invited to see his work. There Joahn met Friedlieb, a student studying under Doebereiner. They immediately became good friends. After looking at Friedlieb extractions on belladonna, Joahn gave Friedlieb a box of mocha Arabian beans to extract. Months later, Friedlieb extracted the world’s first pure caffeine sample which was in a form of white powder. But Friedlieb past away in poverty in 1867, after losing his job when the company disagreed to let him do further instigations
Properties of Caffeine Other names includes:1,3,7-trimethylxanthine, trimethylxanthine, theine, mateine, and guaranine (depending on where it is found) Compound name: Xanthine Alkaloid Molecular Formula: C8H10N4O2 Molar Mass: 194.19 g/mol Appearance: Odorless, white crystals or powder Solubility in H2O: Slightly soluble (depends on temperature) Other Solvents: Alkali benzoate and salicylates Melting point: 237°C Boiling point 178°C
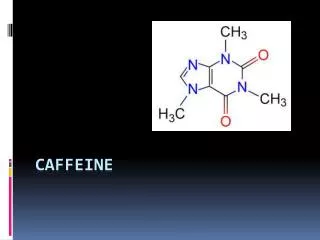
CaffeineStructural Diagram • Caffeine is made of Carbon, Hydrogen, Nitrogen and Oxygen. • When the chemical equation is balanced it forms; • C+H+N+O -> C8 H10 N4 O2 • Also known as 1,3,7-Trimethylpurine-2,6-dione • Caffeine is a covalent bond because the elements that make the molecue are all non-metals.
Decaffeination When extracting of caffeine from coffee commercially, the first step is to clean the green coffee beans with pure water to moisten and prepare for the extraction. Then the beans are put in with a small amount of hot water that contains caffeine and other compounds for flavor. Add in a solvent named trichloroethylene until 97% of the caffeine is removed and trapped by a carbon filter while the flavor still stays in the bean. This step is repeated until the desired amount of decaffeination. Leftover solvent is removed after by steam distillation. The steam also removes the wax from the beans, which are then roasted and ready to be packaged.
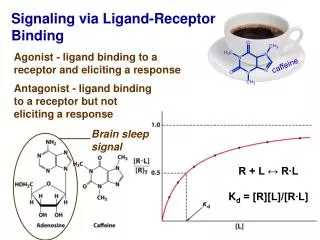
Effects of Caffeine on Human Brains Caffeine has a structure similar to adenosine (a substance slows brain activity and causes tiredness), when it enters the body it attaches to adenosine receptors in the brain. It blocks the receptors, and then caffeine increases the activity in the brain, making the person feel awake or alert. Caffeine is also similar to drugs like heroin, it increases dopamine levels (a chemical in the brain that makes a person feel happy) but not as intensely. It can also become addictive, people will need to take more to get the same effect. When trying to stop using caffeine, they can go through headaches, fatigue, and depression.
Effects of Caffeine and Pharmaceutical uses Caffeine is a central nervous system (brain and spinal cord) stimulant. Caffeine stimulates the central nervous system which increases the feeling of alertness and wakefulness, faster and increased focus, and better body coordination. Used as prescription or non-prescription drugs to reduce fatigue and regain mental alertness for patients when sudden weakness or drowsiness occurs. Used in pain relievers, such as aspirin and acetaminophen to reduce headaches and migraines. Can be injected into premature babies with breathing problems.
Concerns on Caffeine May cause side effects such as: difficulty sleeping, nervousness, trouble breathing or nausea. The “good feeling” can only last for a few hours before returning back to fatigue. Must avoid consuming large amounts of caffeine that contains in foods and beverages, such as coffee, tea, soft drinks, and chocolate with caffeine tablets. Not to be used to replace sleep. It only makes you feel alert but you may not be. Can change blood sugar levels. Diabetics will need to watch closely to their blood sugar while taking caffeine. Caffeine may cause side effects in babies, if mothers who consume caffeine and breast-feed their milk at the same time.
Caffeine Today North America, 90% of adults consume caffeine daily basis. World's main source of caffeine is from the coffee bean (the seed of the plant), where coffee is brewed. Caffeine is mostly known to be in beverages, such as coffee, tea, soft drinks and energy drinks that gives people a quick fix to tiredness. The caffeine in these drinks are most likely from the ingredients used, not from the coffee bean itself. Caffeine can also be found in painkillers, gum, skincare products or even weight loss supplements.
Citations • "The Chemistry of Caffeine - Www.ChemistryIsLife.com.". http://www.chemistryislife.com/the-chemistry-of-caffe • "Basic Prototype Page." Basic Prototype Page. http://www.chm.bris.ac.uk/webprojects2001/tilling/isolation.htm • The History and Use of Caffeine. (n.d.), from https://energy-boosters.knoji.com/the-history-and-use-of-caffeine/ • Caffeine Uses, Effects & Safety Information - Drugs.com. (n.d.). from http://www.drugs.com/caffeine.html • Medical News Today. http://www.medicalnewstoday.com/articles/285194.php?page=3#what_are_the_risks_of_consuming_caffeine. • "The History and Use of Caffeine." https://energy-boosters.knoji.com/the-history-and-use-of-caffeine • "CoffeeForLess.com Learning Center." CoffeeForLess.com. http://www.coffeeforless.com/articles/