Protein-protein interactions
E N D
Presentation Transcript
Protein-protein interactions Masoud Youssefi, MD,PhD Division of microbiology/virology
Introduction • important field in cell biology, biochemistry Localization and trafficking posttranslational modifications signaling networks • also important field in viral replication • very difficult to predict • two main patterns: ■ domain-domain interactions ■domain-peptide interactions
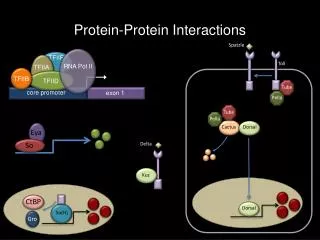
An example: virion assembly • The components come together and the Nucleocapsid is formed which in turn will become completed to the whole particle. • The assembly process begins when concentration of structural proteins is enough within the cell to drive the process. • Many protein-protein, protein-nucleic acid and in case of membrane viruses protein-membrane (fatty acid) interactions are needed.
The mechanism of interaction • Non-covalent so reversible • Van del waals forces • Hydrophobic interactions • Electrostatic bonds • Hydrogen bonds • For strong couplings very accurate force field potentials are needed
Overview of techniques • Gel filtration • Far western blot • Affinity chromatography • Co-immunopercipitation • Capillary elecrophoresis • Biosensor • FRET microscopy • Confocal microscopy • 2 hybrid assay • Protein microarry • Maspec • NMR • Co-crystallization for crystallography
Gel filtration chromatography • Also called ”Size exclusion” • Porous made up of cross-linked polymers • Small molecules are trapped by the beads • For self assembling proteins monomers come later
Far western blot • Also called ”Blot overlay” • Fractionating proteins on SDS-PAGE • Blotting to nitocellulose or PVDF membrane • Overlaying with a solution of the protein of interest • Binding the added protein to an immobilized protein on the membrane • Detection with antibody against the overlaying protein
Co-Immunoprecipitation • Protein A binds to antibodies • Sepharose beads coated with protein A • Specific antibody binds to the protein of interest • The complex is precipitated by binding to the beads via protein A • Proteins are released from beads by boiling • Western blot
Affinity chromatography • In the case of His- tagged proteins • The His-tagged protein binds to nickel or cobalt column • His-tagged protein and it’s associated protein are eluted from the column by adding imidazole
FRET cont • Cyan fluorescence protein (CFP) and yellow fluorescence protein (YFP) are spectral variants of GFP • Plasmid constructs to fuse the proteins of interest to CFP and YFP • Co-transfection of plasmids to the cells • Fixation of the cells and view by confocal microscopy • Disadvantage:False negative results: If the fluorophores are over 200Ǻ apart while the proteins interact with each other, no signal will be observed
Yeast two hybrid assay • Transcription factor, Gal4p, has DNA binding (BD)(aa1-147) and transcriptional activator(AD)(aa768-881) domains • Stimulates transcription at a promoter reconized by Gal4p (upstream activating sequence,UAS) • Lac Z reporter gene encodes beta-galactosidase which produces blue pigment when the colony is grown in a media containing X-Gal • Disadvantage:time consuming!
Mamalian two-hybrid assay • Is analogous to Y2H assay • Plasmids: 1)Gal4pBD-fusion vector 2)VP16AD-fusion vector(viral activator) 3)luciferase reporter plasmid contaning multiple copies of Gal4p binding sites(UAS) • Co-transfection: in the case of interaction, luciferase activity will be detected • Advantage: good for studying mammalian proteins: they may not fold correctly in yeast or they may require post-tranlational modifications for protein interaction
what are biosensors? • Transducer converts physical change(heat, change in charge, light absorbance, mass) into an electrical signal
Confocal microscopy • A good technique to detect intracellular co-localization of proteins • Point scan laser system minimizes overlaps in image (perfect for imaging Co-localization of proteins)
Overview of techniques • Gel filtration • Far western blot • Affinity chromatography • Co-immunopercipitation • Capillary elecrophoresis • FRET microscopy • Confocal microscopy • 2 hybrid assay • Maspec • NMR • Co-crystallization for crystallography