Blood
Blood. Week 6 Dr. Walid Daoud, A. Professor. Blood ____________________________. Blood is a part of extracellular fluid and composed of 2 parts: 1- Fluid part or Plasma (55%). 2- Cellular part (45%): . Red blood corpuscles (erythrocytes). . White blood cells (leukocytes).

Blood
E N D
Presentation Transcript
Blood Week 6 Dr. Walid Daoud, A. Professor
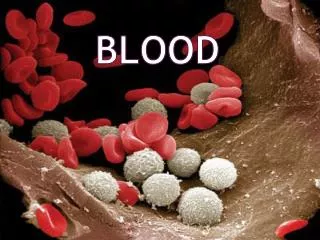
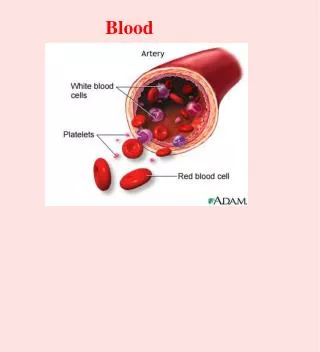
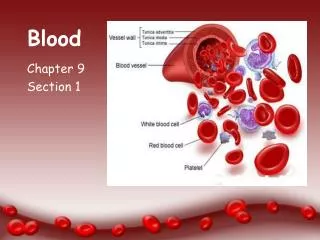
Blood____________________________ Blood is a part of extracellular fluid and composed of 2 parts: 1- Fluid part or Plasma (55%). 2- Cellular part (45%): . Red blood corpuscles (erythrocytes). . White blood cells (leukocytes). . Platelets (thrombocytes).
Plasma____________________________ A yellow clear fluid, 3.5 L, clots on standing. Composition: 1-Water (90%). 2-Organic substances (9.1%): Plasma proteins, lipids, glucose, amino acids, vitamins, enzymes & waste products. 3-Inorganic constituents (0.9%): Na+, Cl-, HCO3-, phosphates and sulphates. 4-Blood gases: O2, CO2 and N2.
Plasma Proteins____________________________ Types: Conc. (g/dl) Mol. Weight 1- Albumin: 3.5-5 69,000 2- Globulins: 2.5 90,000-156,000 3- Fibrinogen: 0.4 340,000 4- Prothrombin: 0.01 68,700 Sites of Formation: 1- Albumin, Fibrinogen, Prothrombin in liver. 2- Globulins: 50% in liver, 50% in plasma cells Albumin/Globulin (A/G) ratio: 1.2-1.6 It decreases in liver and kidney diseases.
Functions of Plasma Proteins__________________________ 1- Osmotic function. 2- Transport function. 3- Defensive function (immunoglobulins). 4- Blood clotting function. 5- Viscosity. 6- Capillary function. 7- Buffer function. 8- Function as a source of tissue proteins.
Red Blood Corpuscles____________________________ - Non-nucleated biconcave discs. - Adult male: 5.4 million / mm3 - Adult female: 4.8 million / mm3 Structure of RBC: - Has a plastic membrane (easily molded). - Contains hemoglobin (Hb). - Contains carbonic anhydrase for CO2 transport. - K+ is the main intracellular cation. - No mitochondria & derives energy from anaerobic metabolism of glucose (glycolysis).
Hemoglobin (Hb)____________________________ - Oxygen-carrying red pigment of RBCs. - Adult male: 15-16 g / dl. - Adult female: 13-14 g / dl - Newborn: 19 g / dl Structure of Hb: 4 subunits: - Each subunit = polypeptide chain + heme - The 4 plypeptide chains are called globin. - Heme is iron protoporphyrin (ferrous iron).
Chemical Reactions of Hb____________________________ 1- With Oxygen: Hb + O2 → Oxyhemoglobin ( HbO2 ) - Each O2 molecule attaches to ferrous iron in heme. - Each Hb molecule can combine with 4 O2 molecules. - Each gram of Hb can combine with 1.33 ml of oxygen to be fully saturated. - O2capacity of 100 ml blood = 16 x 1.33 = 21 ml O2
Chemical Reactions of Hb___________________________ Affinity of hemoglobin to oxygen Causes of increased affinity: - ↓ H+ (alkalis), ↓ temperature, ↓ 2,3-DPG. Causes of decreased affinity: - ↑ H+ (acids), ↑ temperature, ↑ 2,3-DPG.Hydrogen ions and 2,3-DPG compete with O2 to combine with iron of heme part of Hb, so affinity of Hb for O2 decreases.
Chemical Reactions of Hb____________________________ 2- With Carbon Dioxide (CO2): Hb + CO2 → Carbamino compounds CO2 attaches to protein part of Hb.
Chemical Reactions of Hb____________________________ 3- With carbon monoxide (CO): Hb + CO → Carboxyhemoglobin - CO combines with iron and displaces O2 from Hb. - The affinity of Hb for CO is 210 times that for O2, so CO prevents binding of O2 to Hb and ↓ O2 binding capacity of Hb.
Chemical Reactions of Hb____________________________ 4- With strong oxidizing agents: Met-hemoglobin Ferrous iron (Fe2+) in Hb becomes oxidized in presence of strong oxidizing agents to ferric iron (Fe3+), forming met-hemoglobin, which can not combine with oxygen.
Types of Hemoglobin____________________________ 1-Adult hemoglobin (HbA): It contains 2 α and 2 β polypeptide chains and called α2β2 Hb. 2-Fetal hemoglobin (HbF): It contains 2 α and 2 γ chains (α2γ2Hb) HbF has higher affinity to oxygen than HbA which facilitates movement of oxygen from maternal to fetal blood.
Functions of RBCs____________________________ A- Functions of Hb: 1-Transport of O2 from lung to tissues and CO2 from tissues to lung by carbonic anhydrase in RBCs. CO2 + H2O → H2CO3 → H++ HCO-3 2-Buffer H+inside RBCs and can carry CO2 with minimal change in pH. DeoxyHb is a stronger buffer than oxyHb as it forms weaker acids.
Functions of RBCs B-Functions of Membrane: 1-Plastic membrane allows changes in volume of RBCs with minimal change in tension on the membrane. 2-Biconcave shape is suitable for diffusion of gases as it gives maximal surface area 3-Membrane keeps Hb inside RBCs. If HB passes to plasma: -It blocks renal capillaries: renal failure. -Increases blood viscosity & blood pressure. -Increases colloidal osmotic pressure of plasma proteins: increases heart work.
Life Span and Fate of RBCs____________________________ Life span: 120 days. Fate:old RBCs have fragile walls and rupture easily when pass through narrow blood vessels especially in the spleen. Hb is rapidly captured by cells of RES and broken into heme and globin. Heme gives iron and protoporphyrin (bilirubin)
Erythropoiesis____________________________ It is the process of formation of RBCs. Sites: -In fetus: liver (mainly) and spleen. -After birth: bone marrow of long bones. -By age of 20: Bone marrow of long bones can not produce RBCs. -After age of 20: Bone marrow of flat bones.
Stages of differentiation of RBCs____________________________ Pluripotential stem cells in bone marrow can develop in any type of blood cell: Pluripotential stem cells ↓ Colony-forming unit-erythrocyte (CFU-E) ↓ Erythropoietin Erythroblasts ↓ Erythrocytes
Factors Affecting Erythropoiesis____________________________ I -O2 supply to tissues-role of erythropoietin: Tissue hypoxia stimulates production of erythropoietin hormone from kidney & liver which stimulates production of RBCs. e.g, 1- Hemorrhage. 2- High altitude. 3- Athletes. 4- Heart failure. 5- Lung diseases.
Factors Affecting Erythropoiesis____________________________ A- Proteins: Proteins containing essential amino acids are needed for formation of globin part of Hemoglobin (Hb). Decrease dietary protiens leads to failure of Hb formation.
Factors Affecting Erythropoiesis____________________________ B- Vitamins: 1- Vitamin B12 (Cyanocobalamine or Extrinsic or Maturation Factor): Function: Essential fortransformation of mRNA into DNA and nuclear maturation in RBCs. Absorption: Vitamin B12 combines with intrinsic factor in the stomach and absorbed from terminal ileum then passes to blood.
Factors Affecting Erythropoiesis____________________________ Causes of Vitamin B12 deficiency: 1- Absence of intrinsic factor due to atrophy of gastric mucosa (pernicious anemia). 2- Liver diseases. 3- Vitamin B12deficiency in diet (rare). Effects of Vitamin B12 deficiency: 1- Megaloblastic or macrocytic anemia. 2- Decreased number of RBCs in blood. 3- Neurological symptoms.
Factors Affecting Erythropoiesis____________________________ 2-Folic Acid (one of B complex vitamins): Functions: Needed for DNA synthsesis maturation of RBCs. Causes of Folic Acid deficiency: -Dietary deficiency. -GIT diseases interfering with its absorption. Effects of Folic Acid deficiency: Macrocytic anemia.
Factors Affecting Erythropoiesis____________________________ II- Diet: A. Proteins:. B. Vitamins: C- Iron. D- Trace elements.
Factors Affecting Erythropoiesis____________________________ C-Iron: Functions: -Formation of hemoglobin in RBCs and myoglobin in muscles. - As co-factor for oxidation enzymes e.g., catalase, peroxidase, cytochrome oxidase. Total body iron: 4 g. 70% in Hb, 3% in myoglobin, 1% in oxidative enzymes, 26% in liver & spleen.
Factors Affecting Erythropoiesis____________________________ Iron absorption, transport and storage: -Dietary iron (ferric Fe3+) changes to absorbable (ferrous Fe2+) by gastric HCl and vitamin C. -Phytic acid, oxalates, phosphates prevent iron absorption by forming insoluble salts. -Iron is absorbed in upper part of small intestine by active process. In blood iron is carried by plasma protein (transferrin), then iron is transported to bone marrow to form Hb and to muscles to form myoglobin. Excess iron is stored in liver & spleen as ferritin.
Factors Affecting Erythropoiesis____________________________ Causes of iron deficiency anemia: - Decreased iron intake in diet. - Failure of iron absorption. - Chronic blood loss. D- Trace elements: - Cupper: co-factor for synthesis of Hb but does not enter in its formation. - Cobalt: stimulates erythropoiesis and enters in vitamin B12 formation.
Factors Affecting Erythropoiesis____________________________ III- Hormones: 1-thyroxine regulates metabolism. 2-Androgens stimulates erythropoietin. 3-Glucocorticoids increase formation of RBCs IV-Healthy bone marrow: Bone marrow destruction leads to decrease in all types of blood cells. V- Healthy liver: Liver diseases are associated with anemia.