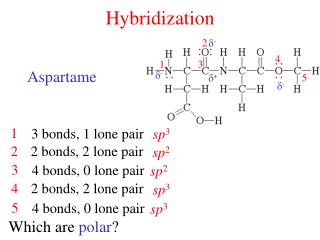
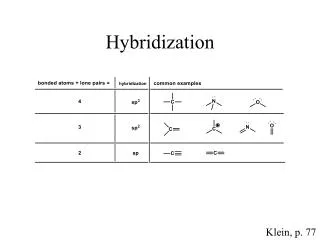
Hybridization
Hybridization. IB Chemistry II Internal Assessment Cook. Hybridization. Atomic oribital overlap to form two types of covalent bonds: Sigma Bonds Pi bonds VSEPR: The shape of the molecule is determined by repulsion between electrons

Hybridization
E N D
Presentation Transcript
Hybridization IB Chemistry II Internal Assessment Cook
Hybridization • Atomic oribital overlap to form two types of covalent bonds: • Sigma Bonds • Pi bonds • VSEPR: • The shape of the molecule is determined by repulsion between electrons • Once we know the lewis structure of a molecule we can predict exactly how the bonds will be orientated with respect to each other in space.
Biochemical Reactions/VSEPR • Lewis structures offer critical features of a substance and biochemical reactions depend on the precise ‘fit’ between the enzyme which controls the rate of reaction and the reacting molecule. • Predictions of molecular shapes are based on valence shell electron pair replusion • This states that electrons pairs found in the outer energy level or valence shell of atoms repel each other and thus position themselves as far apart as possible
VSEPR • The following points will help you apply this theory to predict the shape of molecules: • The repulsion applies to both bonding and non-bonding pairs of electrons • Double and triple bonds are orientated together and so behave in terms of repulsion as a single unit known as a negative charge center. • The total number of charge centers around the central atom determines the geometrical arrangement of electrons • The shapes of molecules is determined by the angles between bonded atoms • Non-bonding pairs of electrons (lone pairs) have higher concentration of charge than a bonding pair because they are not shared between two atoms and so they cause more repulsion than bonding pairs. The repulsion decreases in the following order
How Repulsion decreases • Lone-pair-lone pair>lone pair-bonding pair>bonding pair-bonding pair • It works like this: • Strongest repulsion: Lone pair-lone pair • ` lone pair-bonding pair • Weakest repulsion: bonding pair-bonding pair
Species with 2 negative charge centers • Molecules with 2 with 2 charge centers will position at 180 degrees to each other. The molecule will therefore have a linear shape • Examples • BeCl2 • CO2 • C2H2
Species with 3 negative charges centers • BF3 • HCHO • NO3- • Try this example: • SO2 • First write out lewis structure • How many charge centers do you see? • 1, 2 or 3?
Species with 4 charge centers Molecules with 4 charge centers will position themselves at 109.5 degree giving a tetrahederal CH4 NH4+ Practice drawing lewis structures for the above and indicate the charge centers