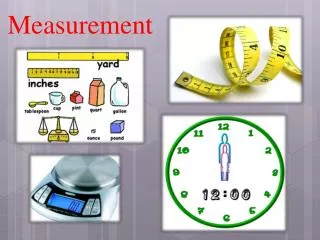
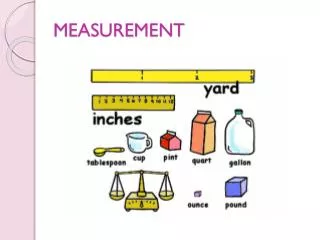
Measurement
Measurement. Measurement In this chapter we will explore the following concepts: 1 . Measurement of a physical parameter 2. Units, systems of units 3. Basic units in mechanics 4. Changing units. Stating a Measurement. In every measurement there is a Number followed by a

Measurement
E N D
Presentation Transcript
Measurement In this chapter we will explore the following concepts: 1. Measurement of a physical parameter 2. Units, systems of units 3. Basic units in mechanics 4. Changing units
Stating a Measurement In every measurement there is a • Number followed by a • Unit from measuring device
Standards of Measurement When we measure, we use a measuring tool to compare some dimension of an object to a standard.
Basic & Derived Quantities • Basic Quantity Must be defined in terms of a standard (meter, kilogram, second). • Derived Quantity Defined in terms of combinations of basic quantities • Unit of speed = meter/second = m/s
International System of Units: In this system the units for the base quantities are: Unit of Length: the Meter A meter is a unit of length, currently defined as the distance light travels within 1/299782458th of a second.
Time Unit –second (s) The time needed for a cesium-133 atom to perform 9,192,631,770 complete oscillations
Volume Unit – the Liter (L) Volume is the amount of space occupied by a substance. S.I unit of volume is liter (L). The milliliter (ml) is commonly used for measuring smaller volumes of fluids. 1 gallon = 4 quarts 1 liter (L) = 1.06 quart 1 quart = 946 mL 1 L = 10 deciliters (dL) = 100 centilliters (cL) = 1000 milliliters (mL)
Mass The mass of an object is a measure of the quantity of material it contains. In the SI system, the mass unit is the kilogram (kg). In the metric system, the mass unit is the gram (g). Compare to US weight units: 1 kg = 2.20 lbs 1 lb = 454 grams
Temperature Scales • The degree of hotness or coldness is called temperature. • The S.I unit of temperature is kelvin • Other Scales: • Celsius (C) scale • Fahrenheit (F) scale • 1 C = 1.8 F
Other Systems of Units • CGS centimeter-gram-second • FPS foot-pound-second
Learning Check From the previous slide, state the tool (s) you would use to measure A. temperature ____________________ B. volume ____________________ ____________________ C. time ____________________ D. weight ____________________
From the previous slide, state the tool (s) you would use to measure A. Temperaturethermometer B.Volumemeasuring cup, graduated cylinder C.Timewatch, clocks D. Weightspring balance
Unit conversions We will work exclusively in SI (MKS) units!
Learning Check What are some S.I units that are used to measure each of the following? A. length B. volume C. weight D. temperature
Some possible answers are A. length inch, foot, yard, mile B. volume cup, teaspoon, gallon, pint, quart C. weight ounce, pound (lb), ton D. temperature °F
Units in the Metric System • length meter m • volume liter L • mass gram g • temperature Celsius °C
Learning Check Identify the measurement in metric units. A. John’s height is 1) 1.5 yards2) 6 feet 3) 2 meters B. The volume of Pepsi in a big bottle is 1) 1 liters 2) 1 quart 3) 2 pints C. The mass of a lemon is 1) 12 ounces2) 145 grams 3) 0.6 pounds
Solution A. John’s height is 3) 2 meters B. The volume of Pepsi in a big bottle is 1) 1 liter C. The mass of a lemon is 2) 145 grams
Units of Volume: • The SI unit of volume is the cubic meter (m)3. • A more convenient unit of volume for everyday use is the liter, a non-SI unit. • A liter (L) is the volume of a cube that is 10 centimeters (10 cm) along each edge (10 cm 10 cm 10 cm = 1000 cm3 = 1 L).
Units and Quantities Scientists commonly use two equivalent units of temperature, the degree Celsius and the kelvin. Units of Temperature
Units and Quantities • The joule (J) is the SI unit of energy. • One calorie (cal) is the quantity of heat that raises the temperature of 1 g of pure water by 1°C.
Section Quiz. 1. Which of the following is not a base SI unit? • gram • meter • second • mole
Section Quiz. • A temperature of 30 degrees Celsius is equivalent to • 303 K. • 300 K. • 243 K. • 247 K.