Hemoglobin and hemoglobinpathies
Hemoglobin and hemoglobinpathies. Srbová Martina Kotaška Karel. Hemoproteins . Consist of hem cyclic tetrapyrrole 1 iron cation Fe 2+ bound in the middle of tetrapyrrole skelet by coordination coavalent bonds conjugated system of double bonds. methine bridge. pyrrole ring.

Hemoglobin and hemoglobinpathies
E N D
Presentation Transcript
Hemoglobin and hemoglobinpathies Srbová Martina Kotaška Karel
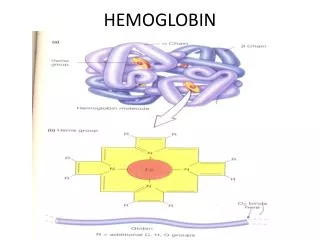
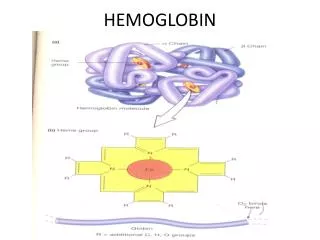
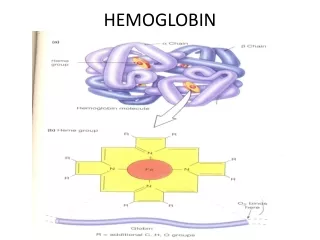
Hemoproteins Consist of hem • cyclic tetrapyrrole • 1 iron cation Fe2+ bound in the middle of tetrapyrrole skelet by coordination coavalent bonds • conjugated system of double bonds methine bridge pyrrole ring
Types of hemoglobin Adult HbA1: 2α and 2β subunits (98%HbA) Adult HbA2: 2α and 2δ subunits (2% HbA) Fetal HbF: 2α and 2γ have higher O2 affinity than HbA – take up oxygen from the maternal circulation Embryoinic: 2and 2 2 and 2 2 and 2 have higher O2 affinity than HbA
Hemoglobin switching Alteration of globin gene expresion during development
Hemoproteins Redox state Fe 2+ • Hemoglobin (transports O2 to the tissues) • Myoglobin (stores O2 in the muscles) • Cytochromes (e- carriers in ETC) • Catalase + peroxidases (decomposition of peroxides) • Cytochrome P-450 (hydroxylation) • Desaturasases FA (desaturation FA) Redox state Fe 2+ Fe 3+
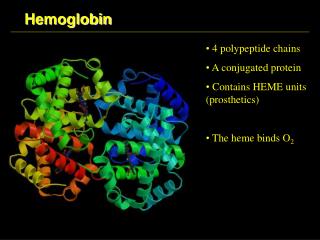
Structure of Hemoglobin • 4 polypetide subunits (globins) • Hb A (adults) heterotetramer 2α a 2β • Each subunit contains 1 hem group • 8 helices (A-H) β subunit • 7 helices α subunit • Hydrofobic pocket - protect hem against oxidation
Structure of Hemoglobin • Hem binding to globin • Fe 2+ is coordinated by N atom from proximal histidin F8 • Binding of O2 • distal histidin E7 hydrogen bonds to the O2
Structureof Hemoglobin • Quaternary structure Interactions between subunits 1) hydrofobic ( between α-β) 2) electrostatic (between α-α; β-β, α-β) • O2 binding – loss of these interactions α1 β2 β1 α2
Structure of Myoglobin • 1 polypeptide chain (153 AA) • 1 hem • Tertiary structures of the α and β subunits are remarkably similar, both to each other and to that of Mb • Skeletal and heart muscles
Binding of O2 (oxygenation) • Oxygenation changes the electronic state of the Fe2+ - hem • Color change of blood from dark purplish (venous) to the brilliant scarlet color (arterial)
Mechanismof oxygen-bindingcooperativity • The binding of the first O2 to Hb enhances the binding futher O2 molecules • O2 affinity of Hb increases with increasing pO2 • Sigmoidal saturation curve • Hyperbolic curve for Mb - no cooperative behavior
Saturation O2 • Hb loads O2 to about 90% saturation under the arterial partial pressure • Hb travels to the tissue where the O2 partial pressure is 20 torr, most of Hb´s bound O2 is released
The diference in oxygen affinity between Mb and Hb is greatest between 5 and 30 torr, where Mb binds much more O2 than does Hb. This difference allows O2 to be released at the tissues from O2 - loaded Hb, and transported to Mb Saturation O2
The loss of electrostatic interactions induce • conformational changes in all other subunits • The movement of Fe 2+ into the heme plane triggers the T→R conformational shift
Conversion of T form→R form T form (tense) R form (relaxed) The binding of the first O2 molecule to subunit of the T-form leads to a local conformational change that weakens association between the subunits R-form
Allosteric effectors Influence the equilibrium between T and R forms • CO2 • H+ • 2,3-bisphosphoglycerate Decrease O2 affinity of Hb
2,3 - bisphosphoglycerate • binds selectively to deoxy-Hb • stabilizes T form • lowers the affinity of Hb for oxygen • oxygen is more readily released in tissues
2,3 - bisphosphoglycerate Clinical aspects: • In people with high-altitude adaptation or smokers the concentration of 2,3-BPG in the blood is increased increases the amount of oxygen that Hb unloads in the capilaries • Fetal hemoglobin (HbF α2γ2), has low BPG affinity – the higher O2 affinity – facilitates the transfer of O2 to the fetus via the placenta
Bohr effect • The binding of protons H+ by Hb lowers its affinity for O2 • Increasing pH, that is, removing protons,stimulates Hb to bind O2 • pH of the blood decreases as it enters tissues because CO2 produced by metabolism is converted to H2CO3 • Dissociation of H2CO3 produces protons • Promote the release of oxygen In the tissues
Bohr effect Oxygen binds to Hb, causing a release protons, which combine with bicarbonate to form H2CO3 Carbonic anhydrase cleaves H2CO3 to H2O and CO2 CO2 is exhaled In the lungs
Hemoglobin determination 1. 2. Direct spectrophotometry of plasma 415 – 460 nm
Derivatives of hemoglobin • Deoxyhemoglobin – Hb without O2 • Oxyhemoglobin – Hb with O2 • Carbaminohemoglobin – Hb with CO2 – CO2 is bound to globin chain – about 15% of CO2 is transported in blood bound to Hb • Carbonylhemoglobin – Hb with CO – CO binds to Fe2+ 200x higher affinity to Fe2+ than O2 – poisoning, smoking
Autooxidation of hemoglobin 3% of hemoglobin undergoes oxidation every day Hem – Fe2+- O2 Hem - Fe3+ + O2•- Methemoglobin reductase reduces methemoglobin FAD, cytochrom b5 a NADH Methemoglobinemia 1. Hereditary deficit of methemoglobin reductase 2. Abnormal hemoglobin HbM (Hb mutation) 3. Exposure to exogenous oxidizing drugs (sulfonamides, aniline) Clinical aspects: cyanosis (10% Hb forms metHb) treatment: administration of methylene blue or ascorbic acid • Methemoglobin– (metHb) contains Fe3+ instead of Fe2+
Glycohemoglobin (HbA1c) • Formed by Hb‘s exposure to high levels of glucose • Nonenzymatic glycation of terminal NH2 group (Val) β-chain • Normally about 5% of Hb is glycated (proportional to blood Glc concentration) • People with DM have more HbA1c than normal ( 5%) • Measurement of blood HbA1c is useful to get information about long-term control of glycemia
Hemoglobinpathies • mutation → abnormal structure of the hemoglobin • Large number of haemoglobin mutations, a fraction has deleterious effects: sickling, change in O2 affinity, heme loss or dissociation of tetramer • hemoglobin M and S, thalassemias 1. Hemoglobin M • Replacement of His E7α by Tyr (Hb Boston) or • Replacement of Val E11β by Glu (Hb Milwaukee) • the iron in the heme group is in the Fe3+ state (methemoglobin) stabilized by the tyrosine or by glutamate • Methemoglobin reductase cannot reduce Fe3+ • methemoglobin can not bind oxygen
2. Thalassemias • Mutation that results in decreased synthesis of α or β-chains • thalassemia mutations provide resistence to malaria in the heterozygous state α- thalassemias – complete gene deletion • 4 α globin genes per cell: • 1 copy of gen is deleted: without symptoms • 2 copies are deleted: RBC are of decreased size (microcytic) and reduced Hb concentration (hypochromic), individual is usually not anemic • 3 copies are deleted: moderately severe microcytic hypochromic anemia with splenomegaly • 4 copies are deleted: hydrops fetalis: fatal in utero Excess β chains form homotetramer HbH which is useless for delivering oxygen to the tissues (high oxygen affinity)
β- thalassemias • β+ – some globin chain synthesis • β0 – no globin chain synthesis Heterozygotes: microcytic hypochromic RBC, mild anemia Homozygotes β0 β0 : severe anemia Excess α chains precipitate in erythroid precursor – their destruction-ineffective erythropoiesis