Hemoglobin
Hemoglobin Blood can carry very little oxygen in solution. Hemoglobin is required to carry oxygen around. Hemoglobin is found in red blood cells Hemoglobin In fact if the body had to depend upon dissolved oxygen in the plasma to supply oxygen to the cells –

Hemoglobin
E N D
Presentation Transcript
Blood can carry very little oxygen in solution. Hemoglobin is required to carry oxygen around. Hemoglobin is found in red blood cells Hemoglobin
In fact if the body had to depend upon dissolved oxygen in the plasma to supply oxygen to the cells – The heart would have to pump 140 liters per minute - instead of 4 liters per minute. Hemoglobin
Each red blood cell can carry about one million molecules of oxygen Hemoglobin is 97% saturated when it leaves the lungs Under resting conditions is it about 75% saturated when it returns. Hemoglobin
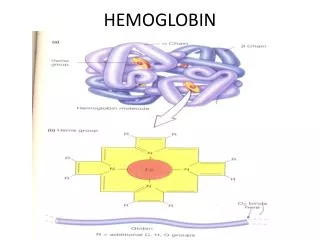
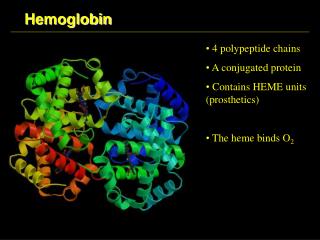
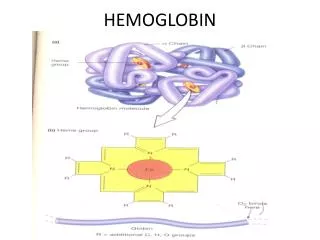
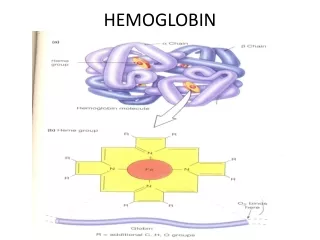
Hemoglobin • Hemoglobin is made from two similar proteins that "stick together". • Both proteins must be present for the hemoglobin to pick up and release oxygen normally. • One of the component proteins is called alpha, the other is beta.
Hemoglobin • Blood cells are made up of two components. • The hemoglobin is in solution inside the cell. • The cell is surrounded by a membrane that holds in the hemoglobin. • A rough analogy would be a rubber water balloon. • The rubber would be the membrane, and the water would be the hemoglobin
At the core of the molecule is porphyrin ring which holds an iron atom. An iron containing porphyrin is termed a heme. This iron atom is the site of oxygen binding. The name hemoglobin is the concatenation of heme and globin Porphyrin Ring
Hemoglobin • Hemoglobin is a remarkable molecular machine that uses motion and small structural changes to regulate its action. • Oxygen binding at the four heme sites in hemoglobin does not happen simultaneously. • Once the first heme binds oxygen, it introduces small changes in the structure of the corresponding protein chain.
Hemoglobin • These changes nudge the neighboring chains into a different shape, making them bind oxygen more easily. • Thus, it is difficult to add the first oxygen molecule, but binding the second, third and fourth oxygen molecules gets progressively easier and easier. • This provides a great advantage in hemoglobin function.
Hemoglobin • When blood is in the lungs, where oxygen is plentiful, oxygen easily binds to the first subunit and then quickly fills up the remaining ones. • Then, as blood circulates through the body, the oxygen level drops while that of carbon dioxide increases.
Hemoglobin • In this environment, hemoglobin releases its bound oxygen. As soon as the first oxygen molecule drops off, the protein starts changing its shape. • This prompts the remaining three oxygens to be quickly released. • In this way, hemoglobin picks up the largest possible load of oxygen in the lungs, and delivers all of it where and when needed.
Hemoglobin The heme group of one subunit, shown in the little circular window, is kept in one place so that you can see how the protein moves around it when oxygen binds. As it binds to the iron atom in the center of the heme, it pulls a histidine amino acid upwards on the bottom side of the heme. This shifts the position of an entire alpha helix, This motion is propagated throughout the protein chain and on to the other chains, ultimately causing the large rocking motion of the two subunits
The mineral, iron, plays an important role in the body’s delivery and use of oxygen to and by working muscles. It binds oxygen to hemoglobin, which then travels in the bloodstream to locations throughout the body. Iron and Hemoglobin
Generally, the more oxygen there is being delivered, the greater the body’s ability to perform work. For this reason, iron receives much attention for its role in supporting aerobic exercise, and it has been postulated that a lack of iron in the body can reduce aerobic capacity and impair endurance performance. Iron and Hemoglobin
Iron deficient red blood cells Low number or cells Note the hollow and blanched appearance of the red blood cells. Iron and Hemoglobin
Sickle Cell Anemia is a genetic disorder that is characterized by the formation of hard, sticky, sickle-shaped red blood cells, in contrast to the biconcave-shaped red blood cells (RBCs) found in “normal” individuals. This disease is caused by a mutation in hemoglobin. Sickle Cell anemia
It consists of two different proteins, an alpha and a beta. If the body doesn't produce enough of either of these two proteins, the red blood cells do not form properly and cannot carry sufficient oxygen. The result is anemia that begins in early childhood and lasts throughout life. Thalassemia
Porphyria is a group of different disorders caused by abnormalities in the chemical steps leading to the production of heme Porphyria
It is characterized by extreme sensitivity to light (exposure to sunlight causes vesicular erythema), reddish-brown urine, reddish-brown teeth, and ulcers which destroy cartilage and bone, causing the deformation of the nose, ears, and fingers. Mental aberrations, such as hysteria, manic-depressive psychosis, and delirium, characterize this condition as well. Porphyria