Introduction to Atoms and Electric Fields
E N D
Presentation Transcript
Introduction to Physical ScienceMonday, Wednesday, ThursdayTom Burbinetomburbine@astro.umass.edu
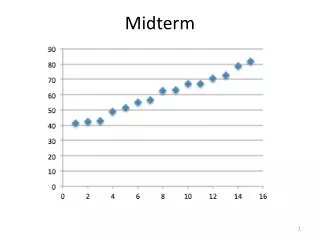
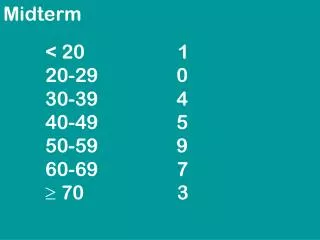
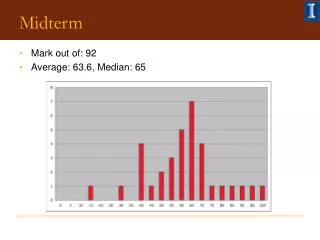
Midterm • Average was 77
Atoms • Composed of : • Electrons – Negative charges • Protons – Positive charges • Neutrons – Neutral charges
Like charges repel • Opposite charges attract
Neutral atom –same number of electrons and protons • If an electron is removed, the atom is positively charged • If an electron is gained, the atom is negatively charged
Fewer electrons than protons, positively charged • More electrons than protons, negatively charged
Conservation of charge – There is no creation or destruction of net electric charge • Electrons can not be created nor destroyed, they can only be transferred from one material to another
Like charges repel • Unlike charges attract
Coulomb’s Law • F = k q1q2 d2 F is force in Newtons q1 is quantity of charge of one particle q2 is quantity of charge of second particle k is 9,000,000,000 N∙m2/C2 (C is a Coulomb)
1 Coulomb is equal in magnitude to the total charge of 6.24 x 1018 electrons • The charge of an electron is approximately −1.602×10−19 C • The charge of a proton is approximately 1.602×10−19 C
Electric Field • Electric field surrounds any charged particle • Electric field decreases with square of the distance from the particle
Electric Potential • A charged particle has potential energy due to its location in an electric field • Electric Potential Energy – Energy possessed by a charged particle due to its location
Electric Potential = electric potential energy amount of charge
1 Volt = 1 joule 1 coulomb
A battery is a device that converts chemical energy directly to electrical energy • Batteries consist of two different metals suspended in an acidic solution.
http://www.green-planet-solar-energy.com/make-a-battery.html