Plant Nutrition
1.2k likes | 3.05k Views
Plant Nutrition. January 2008. Andrew G. Ristvey Wye Research and Education Center. Maryland Cooperative Extension College of Agriculture and Natural Resources University of Maryland. Plant Nutrition. Master Gardener Program. Objectives for this topic include:

Plant Nutrition
E N D
Presentation Transcript
Plant Nutrition January 2008 Andrew G. Ristvey Wye Research and Education Center Maryland Cooperative Extension College of Agriculture and Natural Resources University of Maryland
Plant Nutrition Master Gardener Program Objectives for this topic include: * The essential macro and micronutrients necessary for plant growth and the basic mechanisms for availability and uptake of nutrients. * Organic and inorganic fertilizers and how they are used by the plant. * The negative effects of over-applied or mis-applied fertilizers. * Appropriate timing of fertilizer application and fertilization for special situations
Growth Factors: What do plants need to grow? 1. 2. 3. 4. 5. 6.
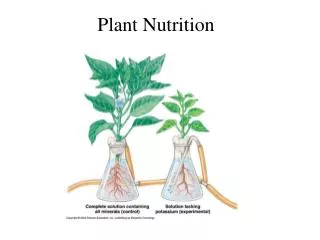
What is an essential plant nutrient? All the nutrients needed to carry out growth and reproductive success; full life cycle The criteria for essentiality: Arnon and Stout, 1939 • Omission of the element will result in abnormal growth • The element cannot be replaced or substituted • The element must exert its effect directly on growth
What is an essential plant nutrient? There are 17 known (accepted) elements that are essential for plant growth Hydrogen, Oxygen, Carbon – plant gets from air and water The other 14 are mineralized elements derived from soil (or air as in N) Other nutrients being studied: Silicon, Cobalt, Aluminum
Relationship between plant growth and nutrient concentration • What happens when a nutrient or nutrients are inadequate in supply? • Can the concentration of a nutrient be too high?
What is an essential plant nutrient? von Liebeg’s ‘Law of the Minimum’ Plant growth progresses to the limit imposed by the nutrient in least supply
What is an essential plant nutrient? von Liebeg’s ‘Law of the Minimum’ Plant growth progresses to the limit imposed by the nutrient in least supply tires chassis engines
Macronutrients Micronutrients
Forms in which nutrients exist • cation – positively charged ion • anion – negatively charged ion • neutral – uncharged • Plants used the mineralized from of a nutrient • It does not matter to the plant where it comes from
ammonium – NH4+ potassium – K+ calcium – Ca+2 magnesium – Mg+2 iron – Fe+2, Fe+3 zinc - Zn+2 manganese Mn+2, Mn+4 copper – Cu+2 cobalt – Co+2 nickel – Ni +2 nitrate – NO3- phosphate – H2PO4- , HPO4-2 sulfate - SO4-2 chlorine – Cl- borate - H3BO3, H2BO3-, B4O7-2 molybdate – MoO4-2 So which nutrients exist in what form? Anions Cations
Factors that affect nutrient uptake • Getting nutrients to the plant roots • Nutrients are water soluble • What factors affect nutrient availability • pH • Cation Exchange Capacity • Colloids (humus, clay)
Getting nutrients to the roots: Mechanisms for nutrient delivery • mass flow • the passive movement of nutrients in soil water to roots • diffusion • the movement of nutrient from regions of high concentration to regions of low concentration • root interception • direct contact of nutrients with roots as roots grow and explore soil
Getting nutrient to the roots: Mechanisms for nutrient delivery
Properties Affecting Nutrient Availability • Chemical Properties - pH p = potential or power H = hydrogen • pH and hydrogen ion concentration are inversely related. • As pH increases, hydrogen ion concentration decreases.
Properties Affecting Nutrient Availability • Chemical Properties - pH • Logarithmic scale pH of 6 has 10x more H+ than pH 7
Properties Affecting Nutrient Availability • Chemical Properties - pH pH affects the availability of nutrients
ammonium – NH4+ potassium – K+ calcium – Ca+2 magnesium – Mg+2 iron – Fe+2, Fe+3 zinc - Zn+2 manganese Mn+2, Mn+4 copper – Cu+2 cobalt – Co+2 nickel – Ni+2 Properties Affecting Nutrient Availability • Chemical Properties – Cation Exchange Capacity C E C Anions Cations • nitrate – NO3- • phosphate – H2PO4-HPO4-2 • sulfate - SO4-2 • chlorine – Cl- • borate - H3BO3, H2BO3-, B4O7-2 • molybdate – MoO4-2
H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ Growing Media - Chemical Properties • Chemical Properties - pH pH affects the availability of nutrients Negatively charged chemical groups OH- on humic particles Sometimes associated with Fe and Al in clays pH High or Low ? OH- OH- OH- Low OH- OH-
H+ H+ H+ H+ H+ H+ H+ Growing Media - Chemical Properties • Chemical Properties - pH pH affects the availability of nutrients Negatively charged chemical groups OH- on humic particles Sometimes associated with Fe and Al in clays pH High or Low ? OH- OH- OH- High OH- OH-
Properties Affecting Nutrient Availability • Chemical Properties – Cation Exchange Capacity C E C The ability of a soil or substrate to provide a nutrient reserve It is all the exchangeable cations the soil or substrate can adsorb The CEC of a soil depends on colloids and pH The higher the CEC of a soil the better buffering capacity
+ Properties Affecting Nutrient Availability • Chemical Properties – Colloids and CEC Colloids - very small particles in soil that are chemically reactive (charged) – humus, clay attracts Fe++ Fe++ Mg++ K+ H+ H+ H+ Ca++ Mg++ Mg++ K+ Mn++ Mn++
Growing Media - Chemical Properties • Chemical Properties - Colloids and CEC pH affects the availability of nutrients Example of one scneario: some nutrients become more available at low pH Mg++ Fe++ Mn++ OH- Ca++ OH- Fe++ Mn++ Fe++ Mn++ OH- Mg++ Fe++ OH- OH- Mn++ Fe++
H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ H+ Growing Media - Chemical Properties • Chemical Properties – CEC pH affects the availability of nutrients H+ ions vie for space, certain ions released becoming available Mn++ Fe++ Mn++ pH ≈ 5.8 OH- Ca++ OH- Fe++ Mn++ OH- Mn++ Fe++ Ca++ OH- OH- Mn++ Mn++ Fe++
Types of Soil Colloids 100-300 humus vermiculite 120-150 60-120 montmorillonite 15-40 illite iron oxides 0-3* Properties Affecting Nutrient Availability • Chemical Properties – Cation Exchange Capacity C E C The ability of a soil or substrate to provide a nutrient reserve • Cation Exchange Capacity • (cmolc/kg of colloid)
What’s on the Bag N P K 10 – 10 – 10 # - # - # – P2O5 – N K2O 1.00 – 0.44 – 0.83 N – P – K
The Major Players – N and P • Nitrogen • NO3- N and NH4+-N or urea • Phosphorus • H2PO4--Pat pH of 5.0 to 6.5
NO3- N and NH4+-N or urea Nitrogen (N) • utilized for a variety of structural and metabolic compounds • over half of N in plants is found in the leaves of plants • between 15 and 30% of that leaf nitrogen goes into the production of Ribulose 1-5-biphosphate carboxylase or Rubisco • Nitrogen is very mobile within the plant
Nitrogen (N) NO3- nitrate • taken up by plants passively and actively • uptake increases pH in soil • best uptake pH range between 4.5 and 6 • nitrate can be stored in plant • nitrates leach
Nitrogen (N) NH4+ ammonium • taken up by plants passively and actively • decreases pH in soil • ammonium (ammonia) cannot be stored • must be assimilated immediately by carbon • ericaceous species utilize
H2PO4- -Pat pH of 5.0 to 6.5 Phosphorus (P) • High pH, P binds with calcium • Low pH P, binds with iron • High P fertilizers do not promote root growth • Utilized for energy transfer, membrane structure, nucleic acids, proteins • Mobile in plant
Nutrient Interactions: Relationships of elemental excess in growing media to potential nutrient deficiencies in plant tissue.
Mobility of Plant Nutrients:Mobility of elements in the plant often defines the location of visual symptoms of nutrient deficiencies or toxicities: * Most recently matured leaves are the most accurate leaf sample for nutrient analysis.
Nutrient Form:Organic or Inorganic? • Plants used the mineralized form of a nutrient • It does not matter to the plant where the nutrient comes from, as all nutrients taken up are in a mineralized form • See handout on types of organic and inorganic fertilizers • However adding composted organic matter to your soil will aid in nutrient availability • See lesson on soils
Nutrient Form:Composts and Teas? • Composts are denatured organic materials • A true aerobic compost requires 3 things • Aeration • Moisture – 40 to 60 % • A C:N ratio of 30 to 1 • Anaerobic composting – less heat, more break down, • increased humus production, but more noxious gases • Making teas from composts is easy, however • making a consistent product is not • Anti-pathogen properties
Foliar Nutrient Application • Plants use the mineralized form of a nutrient • The majority of nutrient uptake are via plant roots • Nutrients can be applied via foliar application • Foliar application should merely be supplemental • For most nutrients • If foliar application is the primary method of nutrition something is wrong with your soil ! (or roots)
Other Negative Effects of Nutrient Over-application • Runoff • Physiological responses • may affect root growth e.g. recent evidence shows P does not promote root growth • may affect flowering e.g. over application of N and other nutrients may stimulate vegetative growth as in grapes • Inappropriate fertilizers • NO3 is not well utilized by ericaceous species • Balance your NO3 with your NH4 • good for most plants
Timing of Fertility • Evidence of periodicity in nutrient uptake in some species • evidence for opposite shoot growth/root uptake periods • fall uptake for spring growth • Lawn care specialists suggest fall fertilization • Arborist stress fall fertilization of trees and shrubs • Tree nursery recommendations stress split fertilization • early spring and mid summer • Some concern over cold hardiness issues with fall N fertility
Fertility - special situations • Drought fertility • Water is the most important growth regulator • No water, no growth regardless nutrients • Fertilizing under drought conditions is not recommended • High EC’s in soil can damage roots • New Plantings • Recent recommendation discourage fertility with new plantings ? What condition (nutrient reserve) were the plants in at purchase • Watering is more important
Suggested Readings Growing Media for Ornamental Plants and Turf. Handrek, K and N. Black. Uni. of New South Wales Press ISBN 0 86840 333 4
13% Plant 8% 15% 1% Pruning Runoff Substrate Plant Uptake Efficiency 21% Where does the Nitrogen go ? Drip 13 g N 63% ? • Both Liquid and CRF holly data, 2001
5% Plant 3% 22% 1% Pruning Runoff Substrate Plant Uptake Efficiency 8% Where does the Nitrogen go ? Overhead Irrigation 33 g N Both CRF and Liquid Feed ? 69% Take home message – great microbial competitiion for N Holly data, 2001
Fertility - special situations • Mycorrhizal Symbiosis • Fungal infection creates a mutualistic relationship with plant • Ectomycorrhizal and Endomycorrhizal (more common) • Very useful to the plant under conditions of low fertility • High fertility retards rate of infection • Fungal mycelia are smaller, have greater surface area than plant roots • Potential disease resistance, drought resistance via symbiosis • Mycorrihzae take C compounds from plant… initially slows growth … eventual long term benefits
N Fertility Recommendations(Turf) • N Fertilizer plan considerations • what types of N should be applied • annual N application rates • application timing
N Fertility Recommendations(Turf) • N Fertilizer - types • All soluble or mixed with slowly available • nitrate, ammonium or both • turf uses mainly nitrate (NO3) • nitrate taken up within 3 days of application • leaching potential high for nitrate • should not use in areas that are leaching prone • should use a 50% WIN formula
N Fertility Turf Recommendations • N Fertilizer – rate issues • how much to apply per application • how much to apply per year • N Fertilizer Recommendations • all soluble – no more than 1 lbs per 1000 sq.ft • nitrate, ammonium or both • can increase rate if you have S.R. N, but only up to the annual max rate
N Fertility Turf Recommendations Table 1. Nitrogen Recommendations for Commercially Maintained Turfgrass on Sites Total Nitrogen Annually (lbs. N/1000 ft2) • adjust if mulching or in low traffic areas
N Fertility Turf Recommendations Table 2. Recommended Periods for N Fertilization of Turf Areas.