10. Amino acids/Proteins Chapter 17
1.27k likes | 1.59k Views
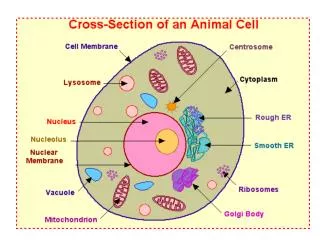
10. Amino acids/Proteins Chapter 17. Protein - More than an Energy Source. Proteins / polypeptides - chains formed by the condensation/combination of 20 different - amino acids. Polypeptides - may be di-, tri -, etc; up to 10 a.a.

10. Amino acids/Proteins Chapter 17
E N D
Presentation Transcript
10. Amino acids/Proteins Chapter 17
Protein - More than an Energy Source Proteins / polypeptides - chains formed by the condensation/combination of 20 different - amino acids. • Polypeptides - may be di-, tri -, etc; up to 10 a.a. • Proteins - longer than 10 a.a. units; ie. MW>10,000
Amino Acids - Protein building blocks An amino acid is a compound having both a carboxyl group(-COOH) and an amino group(-NH2). H H2N C COOH R All amino acids from protein have the -NH2 attached at the C to the –COOH (as well as the H- & R-). All naturally occurring -amino acids, except glycine (R=H), are chiral and the ‘L’ stereoisomer.
H H2N C COOH R There are 20 -amino acids in naturally occurring protein. By convention the -NH2is placed ‘to the left’. Each aa has a ‘common’ name often ending in ‘-ine’. There are ~150 other physiologically important amino acids, GABA (a neurotransmitter).
Amino acids • Contain both an acidic functional group (COOH) and a basic one (-NH2), NH or N • Thus reactions are highly pH dependent
pH dependent properties • Zwitterionic structures contain both N-H+ and COO-. • At low pH, protonate COO-. • At higher pH : lose H on N • Isoelectric pH: differs for each amino acid (due to structural differences)
Leucine ionic forms • Cation below pH 2.4 • Neutral between pH 2.4 and 9.6 • Anionic above pH 9.6
Leucine zwitterion • pH>2.4 • pH < 9.6
Dipeptides • Consider the 2 amino acids glycine (G) and alanine (A). • How many dipeptides can be made if these are randomly mixed? • GG, AA, GA and AG • N terminal on LHS; C terminal on RHS
Tripeptides • Consider amino acids Glycine (G), Alanine (A) and Phenylalanine (P) • How many different tripeptides are possible if each amino acid must be present?
Possible tripeptides • 3 choices for the N-terminal amino acid • 2 choices for middle • 1 choice for the C terminal amino acid • Thus 3 x2 x1 =6 choices if each aa must be present. • But total number possible is 3 x3x3 =27; includes AAA, PPP, GGG etc
Protein Structure • The only unambiguous way to determine the overall structure of any molecule is………….. • Sequence of amino acids can be determined using the enzyme carboxypeptidase (cleaves one aa at a time from the C terminal end)
Levels of Protein Structure Primary structure - the sequence of amino acids inthe peptide chain and the location of the disulfide bridges. Secondary structure - a description of the conformation/ shape of the backbone of the protein. Tertiary structure - a description of the 3D structure of the entire polypeptide. If the protein has more than one chain it can have a quaternary structure.
Some Protein Sequences Oxytocin – contracts smooth muscle (induces ‘labour’) Phe - Gln Tyr Asn Cys Cys- S-S Pro - Arg - Gly Ile - Gln Tyr Asn Cys Cys- S-S Vasopressin - diuretic Pro - Leu - Gly
Val- Glu Gln Ile-Gly Insulin (21 + 30) CysCys-Ser-Leu-Tyr-Gln-Leu-Glu-Asn-Tyr-Cys-Asn Cys-Thr-Ser-Ile Cys-Gly-Ser-His-Leu-Val-Glu-Ala-Leu-Tyr-Leu-Val-Cys-Gly Glu Arg Gly Phe Leu His Gln- Asn-Val-Phe Thr-Lys-Pro-Thr-Tyr-Phe-
Secondary structure of Proteins • Is the fixed arrangement of amino acids resulting from interactions between amide linkages that are close to each other in the protein chain • Interactions can be hydrogen bonds (~ 5 kcal/mol each) • Many H bonds are sufficient to define the shape
Ionic Interactions in Proteins • “salt bridges” • Involve COO- and remote NH3+ groups • Along with H bonding and dispersion forces, these are responsible for the overall shape or “conformation” of the protein
Secondary (20) Structure - sheets H – bond sheets/strands, eg. fingernails, silk
Secondary Structure(20) - the -Helix H-bonding - intramolecular
Tertiary Structure of Proteins • Arises from weaker attractive forces (non polar dispersion forces) between hydrophobic parts of the same chain that are widely separated in the primary structure, but close in space • “intramolecular” • Results in chain twisting and folding
Dispersion forces • Attractive when nuclei are separated by the sum of their van der Waals radii
Tertiary structure of protein: braids and globs • Collagen-a fibrous protein (precursor of gelatin) has a triple helix structure-some elasticity due to interchain interactions • Hemoglobin (a globular protein)
Tertiary Structure (30) - braids & globs collagen hemoglobin
Hemoglobin(H) and Myoglobin (M) • H has 4 polypeptide chains : carries O2, CO2 and H+ in the blood, and possesses quaternary structure • M has a single chain of 153 amino acids: carries O2 from the blood vessels to the muscles and stores it until needed. • Both have Fe II containing heme unit in each chain that binds O2.
To summarize • Myoglobin cannot have quaternary structure since it has only one polypeptide chain • Hemoglobin has 4 polypeptide chains and possesses quaternary structure
Enzyme structure • Many enzymes are proteins and their specific binding properties to a substrate depend on their overall molecular shape or “conformation” Lock and key mechanism for activity
Denaturation - any physical or chemical process that changes the protein structure and makes it incapable of performing its normal function. Whether denaturation is reversible depends on the protein and the extent of denaturation. Examples:·heating egg whites (irreversible) ·‘permanent’ waving of hair (reversible)
Protein Chemistry and your hair • Forces combining to keep hair (a) straight (b) in loose waves or (c) in tight curls are: • Disulfide linkages (part of 10 structure) • Salt bridges • Hydrogen bonds
Protein in Human hair • Keratin (fibrous protein) has the S containing amino acid cystine (14~18%) . • S-S bonds (disulphide linkages) between cystine units give hair its strength by connecting the strands and keeping them aligned
Removing the grey (Grecian Formula) • Active constituent is lead acetate • Reacts with the disulfide links in keratin to produce what black compound? • Also does some structural damage
Animal hair protein composition • Sheep’s wool: also the fibrous protein keratin, but with high glycine & tyrosine content
Perm(?) – have your keratin 1o structure modified HSCH2COOH H2O2
Use some Protein Chemistry on your hair! • Slightly basic solution of thioglycolic acid is used: cleaves the disulfide links and makes new SH bonds (reset hair) • Then Dilute! Peroxide used in final Oxidation step of “perm” (otherwise bleaching effect!) • Covalent S-S bonds in new positions give permanent structure (recall : position of the disulfide linkages is part of 1o structure)
Hydrogen bonding and your hairdo • Hydrogen bonds N-H....O=C Between adjacent strands of fibrous protein are much weaker than the S-S covalent bonds, but there are many more hydrogen bonds, which form a large part of hair structure • Hence excess water will break these up and permit restructuring of hair upon drying • Water not strong enough to break S-S bonds
Hair gels • First ingredient is water • Contain “protein mimics” • Water miscible copolymers with low melting points • Dimethylaminomethacrylate
Protein mimics in hair gels • Y=N , thus an amide ; EO & PO are polymer chains
Protein Denaturation • Heat • Mechanical agitation • pH change • Inorganic salts • polar organic solvents • Soaps and detergents
Heating of protein causes denaturation • Frying eggs • Cooking meat-insoluble collagen protein is converted into soluble gelatin to be used in Jello, gravy, or glue (from horses)
Mechanical Agitation • Beating egg whites-proteins denature at the surface of the air bubbles • Cream of tartar (the dipotassium salt of tartaric acid) is added to beaten egg whites to keep them stiff for mousse and meringue preparation, by raising the pH
Disinfection by denaturation • Ethanol acts via denaturation of bacterial protein • Detergents and soaps disrupt association of protein sidechains of bacterial protein
Protein Denaturation: Origin of Cheese? • Arab merchant carrying milk across the desert in a pouch made from sheep’s stomach • Action of heat caused milk to form a watery liquid and a soft curd with a “pleasing taste” • Rennet containing the enzyme Rennin in the sheep’s stomach caused curd formation
Sour milk , Cheese • Increased amount of lactic acid (from fermentation of lactose by lactobacillus bacteria) causes lower pH • Induces protein denaturation and then coagulation • Casein proteins make up 80% of protein in skim milk • Precipitation of casein by low pH results in curds, essential to cheese making