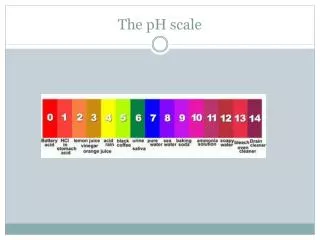
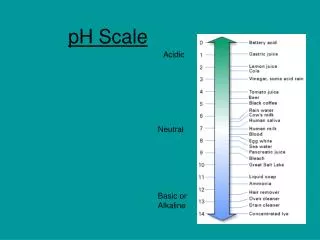
The pH Scale
The pH Scale. Is a substance an acid or a base?. ACIDS. An acid is a compound that increases the number of hydrogen ions when dissolved in water (H+) An acid solution tastes sour An acid can change the color of certain compounds. What is an ion?.

The pH Scale
E N D
Presentation Transcript
The pH Scale Is a substance an acid or a base?
ACIDS • An acid is a compound that increases the number of hydrogen ions when dissolved in water (H+) • An acid solution tastes sour • An acid can change the color of certain compounds
What is an ion? **An ion is an atom that has lost or gained an electron giving it a positive (+) or negative (-) charge. ** Charged Atom
USES OF ACIDS • Citric acid and ascorbic acid (Vitamin C) are found in orange juice • Carbonic acid and phosphoric acid give a “bite” to soft drinks • Acids in your stomach aid in digestion • Sulfuric acid is the most widely used industrial acid in the world. It is used in making metals, paper, paints, and fertilizers.
BASES • A base is any compound that increases the number of hydroxide ions (OH-) when mixed with water • A base solution tastes bitter • A base solution feels slippery • A base can change the color of certain compounds
Uses of Bases • Bases are used in soaps (remember, bases are slippery) • Bases are used to make paper, in oven cleaners, and to unclog drains • Ammonia is a base and is used in many household cleaners
Detecting Acids and Bases • You can detect an acid using an indicator. • INDICATOR- A substance that changes color in the presence of an acid or a base.
INDICATORS • Litmus paper is an indicator • An acid turns blue litmus paper red • A base turns red litmus paperblue
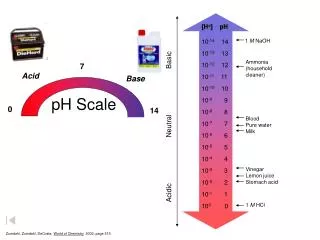
[H+] pH 10-14 14 10-13 13 10-12 12 10-11 11 10-10 10 10-9 9 10-8 8 10-7 7 10-6 6 10-5 5 10-4 4 10-3 3 10-2 2 10-1 1 100 0 1 M NaOH Ammonia (household cleaner) 7 Acid Base 0 14 Blood Pure wate Milk Acidic Neutral Basic Vinegar Lemon juice Stomach acid 1 M HCl pH Scale Zumdahl, Zumdahl, DeCoste, World of Chemistry2002, page 515
pH of Common Substances Timberlake, Chemistry 7th Edition, page 335
Did we Miss something?? • What happens when the pH of a substance is 7? • Ans: A pH level of 7 indicates a Neutral Substance i.e: Water!
Why Learn about pH? • What do you think is the pH level of NYC tap water? • The pH of a swimming pool must be checked periodically. Why? • Is it important for Lakes & Rivers to maintain a certain pH?
How do Antacids Work? • Normal pH in your stomach is 3 • If your stomach pH drops below 3 you don’t feel so well • An antacid neutralizes the acid in your stomach and brings the pH up to a 3 or 4
Antacids • A base can't neutralize your acid all by itself. • A base needs some chemical "helpers” • All antacids contain at least one of these four primary ingredients: • * Sodium • * Calcium • * Magnesium • * Aluminum.
NEUTRAL ACID BASE 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 The pH Scale
NEUTRAL ACID BASE 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 The pH Scale
Let’s see what you’ve learned! • Professor Know-It-All wants to know what you’ve learned. Answer the following questions carefully.
Question #1 • Lemon juice has a pH of 2.2. Is it an acid or a base?
Question #2 • Detergents such as Tide have a pH of about 10. Is Tide an acid or a base?
Question #3 • Seawater has a pH of 8.2. Is seawater an acid or a base?
Question #4 • The inside of your mouth has a pH of 7. Is it an acid or a base?
Question #5 • How do you think we came up with the term “acid rain”?
Question #6 • 409 cleaner contains ammonia that has a pH of 12. What will 409 do to red litmus paper?
Question #7 • A soft drink has a pH of 3.2. What will it do to a piece of red litmus paper?