pH Scale
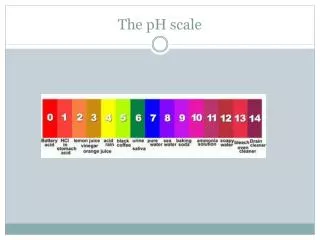
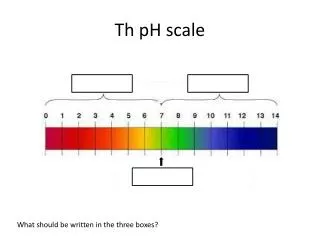
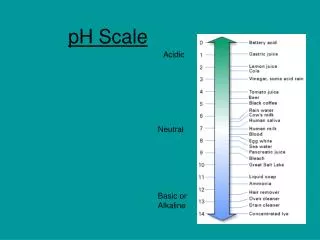
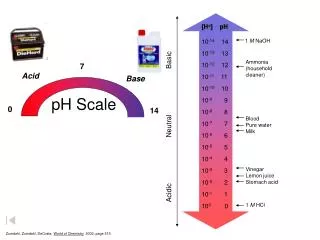
pH. pH Scale. Logarithmic scale. Measures the concentration of hydrogen ions [H + ] in a solution. Range from 0-14. NEUTRAL, pH=7. (pure water) BASE, pH > 7. (ocean water, milk of magnesia, baking sodea ) ACID, pH < 7. (stomach acid/ HCl , vinegar, soft drinks). pH Scale.


pH Scale
E N D
Presentation Transcript
pH Scale • Logarithmic scale. • Measures the concentration of hydrogen ions [H+] in a solution. • Range from 0-14. • NEUTRAL, pH=7. (pure water) • BASE, pH > 7. (ocean water, milk of magnesia, baking sodea) • ACID, pH < 7. (stomach acid/HCl, vinegar, soft drinks)
Let’s take a look at water • 2H2O H3O+ + OH- • [H3O+][OH-]= 1x10-14M2 • [H3O+]= 1x10-7M • [OH-]= 1x10-7M • Kw= 1x10-14M2
Example 1 • An acid is added to water and gives a hydroxide ion concentration [OH-] of 1.0x10-12M. What is the hydrogen ion concentration [H+] ?
How do we measure the pH of a solution? • Acid-base indicators (ex. litmus paper) • pH meter
Equations • pH = -log[H+] • pOH = -log[OH-] • pH + pOH = 14 • [H+][OH-] = 1x10-14 M2
Example 2: What is the pH of a solution with a hydrogen ion concentration of 1x10-4M ? Is this solution acidic or basic ?
Example 3: What is the hydrogen ion concentration of a solution with a pH of 11 ? Is this solution acidic or basic ?
Example 4: Calculate the [H+] and [OH-] of a vinegar solution with a pH of 2.5.
Now, you try…… • 1) [H3O+] = 1.5x10-6M. Find the pH. • 2) A solution’s pH is 3.72. What are the [H3O+] and [OH-] concentrations in the solution?
Thought Question • When an acid is added to a beaker of water, what happens to the [H3O+] and [OH-] in the solution? Why? What happens to the pH? Why?
Acids • Sour Taste • Electrolytes • Identified by the presence of H+ ions (Arrhenius). • Proton (H+) donor (Bronsted-Lowry). • Common Acids: vinegar, citric acid, sulfuric acid, hydrochloric acid.
Bases • Bitter taste • Slippery • Like acids, they are electrolytes • Identified by the presence of OH- ions (Arrhenius). • Proton (H+) acceptor (Bronsted-Lowry).
Strength of Acids and Bases • Based on the concentration of H+ or OH- ions in a solution. • Strong Acids/Bases: completely dissociate into ions in a solution. • Weak Acids/Bases: do NOT completely dissociate into ions in a solution.
HNO3 + NH3 NH4+ + NO3- • Identify the Acid and Base
Stronger the acid, weaker its conjugate base. • Stronger the base, weaker its conjugate acid.
Neutralization ACID + BASE SALT + WATER
Example 1: HBr • Write name
Now you try, HCl • Write name.
Example 2: Hydrofluoric acid • Write chemical formula
Example 3: H2SO4 • Write the name.
Example 4: HClO2 • Write the name.
Try: • HClO3 • HClO • HClO4
Base Nomenclature • Name of cation, name of anion/hydroxide • Ex. NaOH
Tomorrow…. • Briefly brainstorm how we measure the amount of hydrogen ions in a solution? How can we quickly measure the acidity or basicity of a solution?