pH Scale
pH Scale. Ion product constant for water K w. H 2 O H + + OH - K w = [H + ] [OH - ] = 1.0 X 10 -14 mol/L Since [H + ] = [OH - ] for water Then each equals 1.0 X 10 -7 M Water is neutral (neither acid or base).

pH Scale
E N D
Presentation Transcript
Ion product constant for water Kw H2O H+ + OH- Kw = [H+] [OH-] = 1.0 X 10-14 mol/L Since [H+] = [OH-] for water Then each equals 1.0 X 10-7 M Water is neutral (neither acid or base)
The [H+] or [OH-] can be calculated if one is known…. Example: if [H+] = 1.0 X 10-5 Then, [OH-] = 1.0 X 10-9 because [H+] [OH-] must = 1.0 X 10-14
If the [H+] is larger than the [OH-] then the solution is acid If the [OH-] is larger than the [H+], then the solution is basic
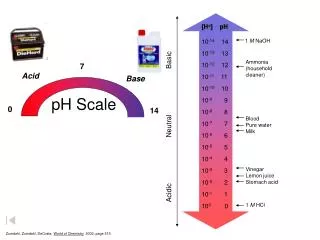
pH Concept that describes how acidic or how basic a solution is… pH = -log [H+] Defined in terms of H+ pH < 7 acid pH > 7 base
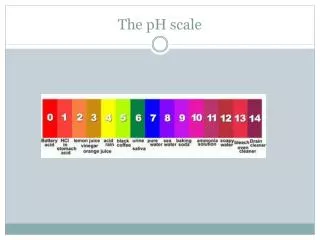
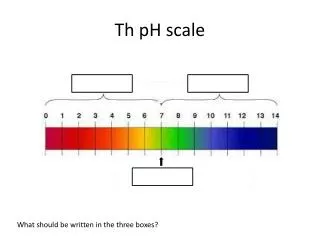
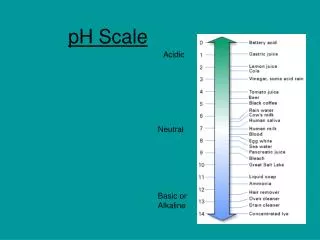
pH scale acidic basic 0 7 14 [H+] = [OH-] 10-7 = 10-7 [H+] > 1.0 X 10-7 [OH-] > 1.0 X 10-7
pOH pOH = -log [OH-] pH + pOH = 14 14 – pOH = pH