pH Scale
pH Scale. 2-27-12 Standard: 5d-students know how to use the pH scale to characterize acid and base solutions. The pH Scale. The pH of a solution is a measure of its hydronium ion concentration.

pH Scale
E N D
Presentation Transcript
pH Scale 2-27-12 Standard: 5d-students know how to use the pH scale to characterize acid and base solutions
The pH Scale • The pH of a solution is a measure of its hydronium ion concentration.
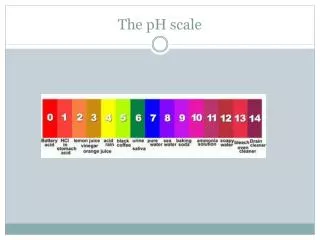
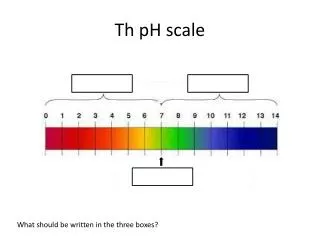
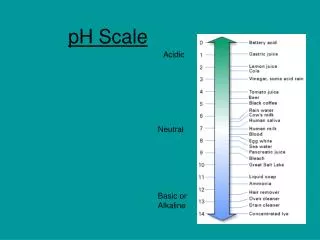
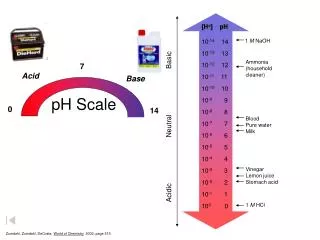
The pH scale is a number scale from 0 to 14 to describe the concentration of hydronium ions in a solution. • A pH of 7 indicates a neutral solution. • Acids have a pH less than 7. • Bases have a pH greater than 7.
If you add an acid to water, the concentration of H3O+increases and the concentration of OH- decreases. • The lower the pH value, the greater the H3O+ion concentration in solution is.
If you add a base to water, the concentration of OH- increases and the concentration of H3O+decreases. • The higher the pH value, the lower the H3O+ion concentration is.
Example • Substance A has a pH of 3. Substance B has a pH of 10. • Is the substance A considered acidic or basic? Is substance B considered acidic of basic? • From this does this mean there are more hydronium ions present or hydroxide ions present for substance A? Substance B? • From Friday does this mean your substances are weak or strong acids or bases?
Try it on your own • Substance A has a pH of 5, Substance B has a pH of 9. • Is substance A and B considered acidic of basic? • From this does this mean there are more hydronium ions present or hydroxide ions present for substance A? Substance B? • Are substance A and B weak or strong acids or bases?
Exit Question • a) What does the pH scale measure? • Given the following pH numbers is the sample a base, acid, or neutral? • A) 6 • B) 12 • C) 7 • Please include a reflection on how you feel about your activity today.