Chapter 22 Transition Elements
Chapter 22 Transition Elements. Valdosta State University. Transition Elements – d- and f-block. Used in construction and manufacturing (iron), coins (nickel, copper, zinc), ornamental (gold, silver, platinum). Densest elements (osmium d=22.49 g/cm 3 , iridium d=22.41g/cm 3 ).

Chapter 22 Transition Elements
E N D
Presentation Transcript
Chapter 22Transition Elements Valdosta State University
Transition Elements – d- and f-block • Used in construction and manufacturing (iron), coins (nickel, copper, zinc), ornamental (gold, silver, platinum). • Densest elements (osmium d=22.49 g/cm3, iridium d=22.41g/cm3). • Highest melting point (tungsten, mp=3410oC) and lowest melting point (mercury, mp=-38.9oC).
Metal Chemistry • Radioactive elements with atomic number less than 83 (technetium 43; promethium 61). • All elements are solids, but mercury. • Have metallic sheen, conduct electricity and heat. • Are oxidized and form ionic compounds. • Some are essential to living organisms: Cobalt (vitamin B12), iron (hemoglobin and myoglobin), molybdenium and iron (nitrogenase). • Compounds are highly colored and used as pigments: Fe4[Fe(CN)6)3 14 H2O (prussian blue), TiO2 (white). • Ions give color to gemstons: Iron(II) ions give yellow color in citrine and chromium(III) ions produce the red color of a ruby. Valdosta State University
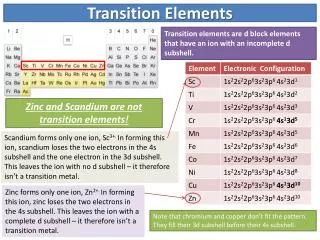
Electron Configurations • General: • [noble gas core] nsa (n-1) db • Valance electrons for transition elements reside in the ns and (n-1) d subshells.
Reactions • All metals undergo oxidation with oxygen, halogens, aqueous acids. • First the outermost electron is removed, followed by one or more d electrons. • Some generate cations with unpaired electrons = paramagnetism. • Are colored. • For first transition series common oxidation numbers are +2 and +3. Fe: [Ar]3d64s2 Fe2O3 Fe3+ [Ar]3d5 Fe + O2 Fe + Cl2 Fe + HCl FeCl3 Fe3+ [Ar]3d5 Fe + Cl2 FeCl2 + H2 Fe2+ [Ar]3d6 Fe + HCl Fe + O2
Most common Trends: Oxidation number Valdosta State University
Trends: Atom Radius Valdosta State University
Trends: Density Valdosta State University
Metallurgy: Element Sources Valdosta State University
Pyrometallurgy • Involves high temperature, such as Fe • C and CO used as reducing agents in a blast furnace • Fe2O3 + 3 C ---> 2 Fe + 3 CO • Fe2O3 + 3 CO ---> 2 Fe + 3 CO2 • Lime added to remove impurities, chiefly SiO2 SiO2 + CaO ---> CaSiO3 • Product is impure cast iron or pig iron Valdosta State University
Hydrometallurgy • Use aqueous solutions (flotation). Some use bacteria. • Add CuCl2(aq) to ore such as CuFeS2 (chalcopyrite)CuFeS2(s) + 3 CuCl2(aq) --> 4 CuCl(s) + FeCl2(aq) + 2 S(s) • Dissolve CuCl with xs NaClCuCl(s) + Cl-(aq) --> [CuCl2]- • Cu(I) disproportionates to Cu metal2 [CuCl2]- --> Cu(s) + CuCl2 (aq) + 2 Cl- Native copper Azurite, 2CuCO3•Cu(OH)2 Valdosta State University
•• H H N H •• H O H •• Coordination Compounds • combination of two or more atoms, ions, or molecules where a bond is formed by sharing a pair of electrons originally associated with only one of the compounds. Valdosta State University
Coordination Chemistry Pt(NH3)2Cl2 “Cisplatin” - a cancer chemotherapy agent Co(H2O)62+ Cu(NH3)42+
Coordination Chemistry An iron-porphyrin, the basic unit of hemoglobin Valdosta State University
Myoglobin / Hemoglobin p.1084
Co atom Coordination Chemistry Vitamin B12 A naturally occurring cobalt-based compound Valdosta State University
Coordination Chemistry • Biological nitrogen fixation contributes about half of total nitrogen input to global agriculture, remainder from Haber process. • To produce the H2 for the Haber process consumes about 1% of the world’s total energy. • A similar process requiring only atmospheric T and P is carried out by N-fixing bacteria, many of which live in symbiotic association with legumes. • N-fixing bacteria use the enzyme nitrogenase — transforms N2 into NH3. • Nitrogenase consists of 2 metalloproteins: one with Fe and the other with Fe and Mo. Valdosta State University
Coordination Chemistry Nickel ion: coordination compounds
Nomenclature • [Ni(NH3)6]2+ • A Ni2+ ion surrounded by 6, neutral NH3 ligands • Gives coordination complex ion with 2+ charge. Ligand: monodentate Coordinate to the metal via a single Lewis base atom.
Inner coordination sphere Nomenclature + Ligand: polydentate also chelating ligands Coordinate with more than one donor atom. (Bidentate) Cl- Co3+ + 2 Cl- + 2 neutral ethylenediamine molecules Cis-dichlorobis(ethylenediamine)cobalt(II) chloride
Bipyridine (bipy) Acetylacetone (acac) Oxalate (ox) Ethylenediamine (en) Bidentate Ligands
Bidentate Ligands Acetylacetonate Complexes Commonly called the “acac” ligand. Forms complexes with all transition elements.
Multidentate Ligands EDTA4- - ethylenediaminetetraacetate ion Multidentate ligands are sometimes called CHELATING ligands
Multidentate Ligands Co2+ complex of EDTA4-
Give the formula of a coordination compound A Co3+ ion bound to one Cl- ion, one ammonia molecule, and two ethylenediamine (en) molecules. • Determine the net charge (sum the charges of the various components). • Place the formula in brackets and the net charge attached. [Co(H2NCH2CH2NH2)2(NH3)Cl]2+
Determine the metal’s oxidation number and coordination number Pt(NH3)2(C2O4) Oxalate: (C2O4)2- Ammonia: NH3 Pt must be 2+ (oxidation number = +2) Coordination number = 4 (two from oxalate and each ammonia filling one). [Co(NH3)5Cl]SO4 Chloride: Cl- Sulfate: SO42- Overall complex must be 2+ Co must be 3+ (oxidation number = +3) Coordination number = 6 (sulfate is not coordinated to the metal).
Nomenclature 1. Positive ions named first 2. Ligand names arranged alphabetically 3. Prefixes -- di, tri, tetra for simple ligands bis, tris, tetrakis for complex ligands 4. If M is in cation, name of metal is used 5. If M is in anion, then use suffix -ate CuCl42- = tetrachlorocuprate 6. Oxidation no. of metal ion indicated in roman numerals. Cis-dichlorobis(ethylenediamine)cobalt(III) chloride
Nomenclature Co(H2O)62+ Hexaaquacobalt(II) Cu(NH3)42+ H2O as a ligand is aqua Tetraamminecopper(II) diamminedichloroplatinum(II) Pt(NH3)2Cl2 NH3 as a ligand is ammine Valdosta State University
Nomenclature Tris(ethylenediamine)nickel(II) IrCl(CO)(PPh3)2 [Ni(NH2C2H4NH2)3]2+ Vaska’s compound Carbonylchlorobis(triphenylphosphine)iridium(I)
Geometry of Coordination Compounds Defined by the arrangement of donor atoms of ligands around the central metal ion. Valdosta State University
Isomerim of Coordination Compounds • Two forms of isomerism • Constitutional • Stereoisomerism • Constitutional • Same empirical formula but different atom-to-atom connections • Stereoisomerism • Same atom-to-atom connections but different arrangement in space. Geometric and Optical
Constitutional Isomers Aldehydes & ketones 3C, 1O, 6H • Coordination isomerism: it is possible to exchange a ligand and the uncoordinated counterion. • Example: [Co(NH3)5Br]SO4 and [Co(NH3)5SO4]Br • (violet)(red) • Linkage isomerism: it is possible to attach a ligand to the metal through different atoms. • Usually: SCN- and NO2-
sunlight Constitutional Isomers Such a transformation could be used as an energy storage device. Pentaamminenitritocobalt(III) Pentaamminenitrocobalt(III) Valdosta State University
cis trans Stereoisomerism • One form is commonly called geometric isomerism or cis-trans isomerism. Occurs often with square planar complexes. Note: there are VERY few tetrahedral complexes. Would not have geometric isomers.
Geometric Isomers Cis and trans-dichlorobis(ethylenediamine)cobalt(II) chloride
Geometric Isomers For octahedral complexes (MX3Y3): fac isomer has three identical ligands lying at the corners of a triangular face of octahedron (fac=facial). mer isomer ligands follow a meridian (mer=meridional). fac isomer mer isomer
Stereoisomers • Enantiomers: stereoisomers that have a non-superimposable mirror image. • Diastereoisomers: stereoisomers that do not have a non-superimposable mirror image (cis-trans isomers). • Asymmetric: lacking in symmetry—will have a non-superimposable mirror image. • Chiral: an asymmetric molecule. Valdosta State University
Enantiomers [Co(NH2C2H4NH2)3]2+
Stereoisomers [Co(en)(NH3)2(H2O)Cl]2+ These two isomers have a plane of symmetry. Not chiral. These two are asymmetric. Have non-superimposable mirror images.
Stereoisomers [Co(en)(NH3)2(H2O)Cl]2+ These are non-superimposable mirror images: enantiomers
Bonding in Coordination Compounds • Model must explain • Basic bonding between M and ligand • Color and color changes • Magnetic behavior • Structure • Two models available • Molecular orbital • Electrostatic crystal field theory • Combination of the two ---> ligand field theory Valdosta State University
Bonding • As ligands L approach the metal ion M+, • L/M+ orbital overlap occurs • L/M+ electron repulsion occurs • Crystal field theory focuses on the latter, while MO theory takes both into account
energy e g 2 2 2 d(x -y ) dz D0 t 2g d d d xy xz yz Ligand Field Theory All electrons have the same energy in the free ion • Consider what happens as 6 ligands approach an Fe3+ ion: Orbitals split into two groups as the ligands approach. Value of ligand field sppliting: ∆o depends on L: e.g., CN- > H2O > Cl-
e energy d d d xy xz yz t 2 2 2 2 d(x -y ) dz Crystal Field Theory • Tetrahedral ligand field. • Note that ∆t = 4/9 ∆o and so ∆t is small. • Therefore, tetrahedral complexes tend to absorb “red wavelengths” and be colored blue. Dt
Ways to Distribute Electrons • For 4 to 7 d electrons in octahedral complexes, there are two ways to distribute the electrons. • High spin — maximum number of unpaired e- • Low spin — minimum number of unpaired e- • Depends size of ∆o and P, the pairing energy. • P = energy required to create e- pair.
e e n n e e r r g g y y e e g g 2 2 2 2 2 2 d d ( ( x x - - y y ) ) d d z z D E s m a l l t t 2 2 g g d d d d d d x x y y x x z z y y z z e n e r g y e g 2 2 2 d ( x - y ) d z D E l a r g e t 2 g d d d x y x z y z Magnetic Properties of Fe2+ • High spin • Weak ligand field strength and/or lower Mn+ charge • D0 is smaller than P • [Fe(H2O)6]2+ Paramagnetic • Low spin • Stronger ligand field strength and/or higher Mn+ charge • D0 is larger than P • [Fe(CN)6]4- Diamagnetic
High and Low Spin Octahedral Complexes High or low spin octahedral complexes only possible for d4, d5, d6, and d7 configurations.