Understanding Electromagnetic Radiation and Quantum Mechanics
This lecture explores the fundamentals of electromagnetic radiation, including waves that transmit energy, the characteristics of electric and magnetic fields, and the visible spectrum. It covers classical physics failures in explaining phenomena like blackbody radiation and introduces quantum theory concepts such as discrete energy levels and the photoelectric effect. Students will examine particle-wave duality, Heisenberg's uncertainty principle, and Schrödinger's wavefunction in relation to atomic behavior, providing a comprehensive overview of light's nature and atomic structure.

Understanding Electromagnetic Radiation and Quantum Mechanics
E N D
Presentation Transcript
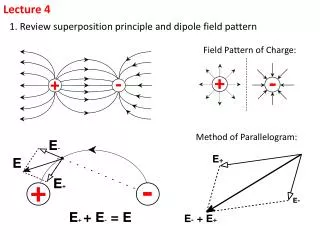
Lecture 4 Understanding Electromagnetic Radiation 1.5-1.7 30-Aug Assigned HW 1.1, 1.16, 1.20 1.21, 1.22, 1.23, 1.24, 1.33, 1.36 Due: Monday 6-Sept
Review 1.2-1.4 • Waves transmit energy • Electromagnetic Radiation • Electric Field • Magnetic Field • The visible spectrum is a VERY small part of the EM spectrum • Sunlight is considered a continuous spectrum • Contains many wavelengths • Sodium and Hydrogen emission spectra are both examples of discontinuous • Hydrogen spectrum • Balmer Series visible • Lyman series UV • Classical Physics cannot explain these observations • Blackbody Radiation • Stefan-Boltzmann and Wien’s laws • Quantum Theory – Discreet energy levels
Emission Lines and Energy Levels • We add energy (heat) to an element • ‘Excited’ electron • When the energy source is removed • Relaxes to ground state (n=1) • What happens when the energy added is more than the n=1 n=∞ transition?
Photoelectric Effect – the QT test • When light (hν) strikes a metal surface, electrons are ejected • Emission ONLY occurs when the incident light exceeds a threshold (φ) • The number of electrons emitted depends on the intensity of the incident light. • The kinetic energies of emitted electrons depend on the frequency of the light. Energy of Incident light Kinetic Energy Of ejected Electron Energy Required For Ejection Video
Photoelectric Effect – the QT test Example: The wavelength of light needed to eject an electron from hydrogen is 91.2 nm. Calculate the velocity of the particle ejected when 80.0 nm light is shone on a sample of hydrogen.
Photoelectric Effect – multiple samples These experiments verify that for each element, discreet amounts of energy are required to eject an electron, φ, which corresponds to a difference between two well defined energy levels
How Do We Find the Work Function? • Calculate the work function for a hydrogen atom.
Particle-like Properties of Photons • What have we talked about that suggests that electromagnetic radiation behaves very much like particles? Photon Hint: Requires a physical interaction between the photon and another particle This other particle is released with a specific hν
Wave-like Properties of Photons • Several observations indicate that photons have intrinsic wave-like properties • Dispersion through a spectrum
Wave-like Properties of Photons • Additional evidence of wave-like properties comes from observing the diffraction patterns of photons
Diffraction and Interference • When two waves traveling along different paths interact, they combine in an additive way. We call this interference. • Constructive Interference - • Destructive Interference –
Diffraction and Interference • Let’s revisit the diffraction patterns: • Where is the constructive interference? • Destructive?
Other uses of Diffraction • Solving the structure of proteins! • Dr. Hurlbert research
Particle-Wave Duality of Photons • Photoelectric Effect tells us that photons act like particles while diffraction and dispersion indicate wave-like properties • We conclude that a photon can take on characteristics of BOTH • Louis de Broglie suggested that all particles should have wavelike properties (‘matter-wave’) Linear momentum (kg m s-1) 1937 Nobel Prize was awarded for this discovery
Repercussions of Particle-Wave Duality • Classical Physics • BUT…if a particle is really a wave, can we know where it is?
Repercussions of Particle-Wave Duality • Model of the atom….electrons • Classical physics predicts a trajectory with known ρ and x. • Particle-wave duality tells us this is NOT the case Classical Interpretation Quantum Mechanical Interpretation
Repercussions: Heisenburg Uncertainty • Niels Bohr and Werner Heisenberg investigated just how precisely we can determine the behavior of subatomic particles. • Two variable MUST be measured for a full understanding • ρ • x • They concluded that there must always be uncertainties in the measurement
Heisenburg Uncertainty Principle • When the position of the particle is well defined, the momentum is not • If the momentum is accurately known, the position is not
Using the Principle • An electron is traveling at 2.05 ± 0.03 x 106 ms-1. With what precision can we simultaneously measure the position of the electron?
Wavefunctions and Schrödinger • We need to adjust the mathematical definition of matter to account for particle-wave duality. • Edwin Schrödinger’s approach was to replace the trajectory of a particle with a wavefunction, ψ. • A wavefunction is: Yes, I rock the bowtie like a champ! Schrödinger also had a cat.
Wavefunctions and Probability • Physical usefulness of ψ came from Max Born • The Born interpretation states that: Probability density Why is this value never negative? What would a negative value indicate?
Wavefunctions and Probability Wake up and insert answer • Ψ2 is a density – dependent on • How do we calculate mass from density? ρ=m/V • Same for ψ2 Example: If ψ2 = 0.5 pm-3, calculate the probability of finding a particle in a sphere with a radius of 1pm.
Schrödinger’s Great Contribution which is often expressed as but what does this mean?
Particle in a Box • Imagine a particle confined to a box with a length of L. • Wavelengths are restricted to those with nodes at 0 and L. Why?
Particle in a Box • How do we find the energy of the particle? Allowed wavelengths are:
Quantization of Energy • We’ve established that for a particle in a 2D box. • Only certain wavelengths fit into the box, n is restricted to integers. • What does this tell us about the levels of energy? • Think about the Photoelectric Effect. Does this make sense?
Changing the Box Length • If we confine the particle to a smaller or larger space. What influence will it have on the energy levels? • Qualitatively • Quantitavely 5 nm box vs. 500 nm box. Calculate n = 1 and n = 2.
Changing the Box Length • If we confine the particle to a smaller or larger space. What influence will it have on the energy levels?
Sample Problems • Use the particle-in-a-box model to calculate the wavelength of the third quanta of a box with a length of 100pm.
Sample Problems • Calculate the probability density for the particle in a box model. Much easier than it looks, isn’t it?
Sample Problems • Derive an equation that allows the difference between two energy levels to be determined.