S N 1 Reactions
450 likes | 844 Views
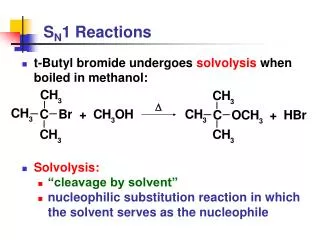
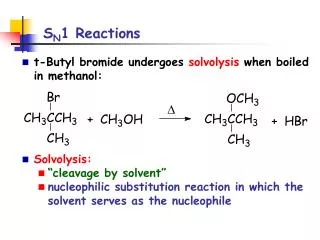
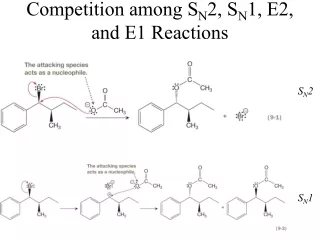
D. S N 1 Reactions. t-Butyl bromide undergoes solvolysis when boiled in methanol: Solvolysis: “cleavage by solvent” nucleophilic substitution reaction in which the solvent serves as the nucleophile. S N 1 Reactions.

S N 1 Reactions
E N D
Presentation Transcript
D SN1 Reactions • t-Butyl bromide undergoes solvolysis when boiled in methanol: • Solvolysis: • “cleavage by solvent” • nucleophilic substitution reaction in which the solvent serves as the nucleophile
SN1 Reactions • The reaction between t-BuBr and methanol does NOT occur via an SN2 mechanism because: • t-BuBr: • too hindered to be SN2 substrate • CH3OH: • weak nucleophile • Solvolysis reactions occur via an SN1 mechanism:
SN1 Reactions • SN1 Reactions • substitution nucleophilic unimolecular • Rate = k[R-X] • 1st order overall • 1st order in [R-X] • zero order in [Nuc] • Only R-X is present in the transition state for the rate determining step • Nucleophile is NOT present in RDS
SN1 Reactions • General Mechanism: • Step 1: • R X R+ + X- • Step 2: • R+ + Nuc - R Nuc Rate determining step
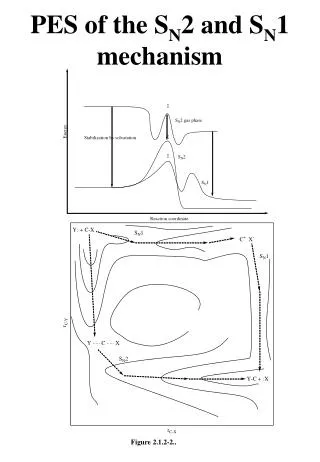
SN1 Reactions Reaction Energy Diagram for SN1 Reactions: Formation of carbonium ion is highly endothermic According to Hammond’s Postulate, the transition state most closely resembles the carbonium ion.
+ + SN1 Reactions • The reactivity of a substrate in an SN1 reaction depends on the stability of the carbonium ion formed: 3o > 2o > 1o > methyl • Allylic and benzylic halides undergo SN1 reactions because the resulting carbonium ions are resonance stabilized.
SN1 Reactions • SN1 reactions involve: • weak nucleophile • H2O not OH- • CH3OH not CH3O- • Substrates that form stable carbonium ion intermediates: • 3o, benzylic, or allylic halide are most favored • 2o (sometimes)
SN1 Reactions Example: Draw the mechanism for the SN1 reaction of t-butyl bromide with methanol • Step 1:Slow ionization of R-X to form carbonium ion: • rate determining step
SN1 Reactions • Step 2: • Fast attack of nucleophile on the carbonium ion
SN1 Reactions • Step 3: (Not needed for all SN1 reactions) • Solvent molecule removes proton, leaving the neutral product
SN1 Reactions-Stereochemistry • The carbonium ion intermediate formed during an SN1 reaction is sp2 hybridized and planar. • The nucleophile can attack from either side of the carbonium ion. • A mixture of both possible enantiomers forms. • Racemization: • a process that gives both enantiomers of the product • not necessarily in equal amounts
+ SN1 Reactions-Stereochemistry Attack from top - H+ - H+ Attack from bottom
SN1 Reactions-Stereochemistry • When the nucleophile attacks from the side where the leaving group was originally, retention of configuration occurs. Attack from top (R) (R)
SN1 Reactions-Stereochemistry • When the nucleophile attacks from the back side (opposite to the original leaving group), inversion of configuration occurs. Attack from bottom (S) (R)
SN1 Reactions-Stereochemistry • For most SN1 reactions, the leaving group partially blocks the front side of the carbonium ion • more inversion of configuration • less retention of configuration
SN1 Reactions-Rearrangements • Carbonium ions often undergo rearrangements, forming more stable cations. • Structural changes resulting in a new bonding sequence within the molecule • The driving force for a rearrangement is the formation of a more stable intermediate. • 1o or 2o carbonium ion rearranges to a more stable 3o carbonium ion or resonance-stabilized carbonium ion
CH3CH2OH + D SN1 Reactions-Rearrangements • A mixture of products often forms as a result of rearrangements during SN1 reactions. • NOTE: Rearrangements cannot occur during SN2 reactions since an intermediate is not formed. Rearranged product Rearrangement occurs via hydride shift.
SN1 Reactions-Rearrangements • Common rearrangements: • Hydride shift (~H) • the movement of a hydrogen atom and its bonding pair of electrons • Methyl shift (~CH3) • the movement of a methyl group and its bonding pair of electrons
SN1 Reactions-Rearrangements • Hydride Shift Mechanism: • Step 1: Formation of carbonium ion and rearrangement:
SN1 Reactions-Rearrangements • Hydride Shift Mechanism: • Step 2: Nucleophile attack and loss of proton (if needed)
EtOH D EtOH D SN1 Reactions-Rearrangements • Example of a Methyl Shift (~CH3):
SN1 Reactions-Rearrangements • Mechanism of ~CH3: • Step 1: Simultaneous (often) shift of methyl group and loss of leaving group:
SN1 Reactions-Rearrangements • Mechanism of ~CH3: • Step 2: Attack of nucleophile and loss of proton (if needed)
SN1 Reactions-Rearrangements Example: Propose a mechanism for the following reaction. CH2CH3OH + D
SN1 vs. SN2 SN2 SN1 • Strong nucleophile • Primary or methyl halide • Polar aprotic solvents (acetone, CH3CN, DMF) • Inversion at chiral carbon • No rearrangements • Weak nucleophile (may also be solvent) • Tertiary,allylic, benzylic halides • Polar protic solvent (alcohols, water) • Racemization of optically active compound • Rearranged products
Na+ -OCH3 CH3OH + Br - E1 Reactions • An elimination reaction involves the loss of two atoms or groups from a substrate, usually forming a new p bond. • Elimination reactions can occur via a first order (E1) or a second order (E2) process.
E1 Reactions • E1 reactions: • Elimination, unimolecular • 1st order kinetics • Rate = k[R-X] • RDS transition state involves a single molecule • General conditions: • 3o and 2o halides • weak bases
E1 Reactions • E1 Mechanism:
D E1 Reactions • E1 reactions almost always occur together with SN1 reactions. E1 SN1
E1 Reactions + CH3CH2-O-H H
E1 Reactions • Once formed, a carbonium ion can: • recombine with the leaving group • react with a nucleophile forming a substitution product (SN1) • lose a proton to form an alkene (E1) • rearrange to form a more stable carbonium ion and then: • react with nucleophile • lose a proton to form an alkene
E2 Reactions • E2 reactions: • Elimination, bimolecular • 2nd order kinetics • Rate = k[R-X][B-] • RDS transition state involves two molecules • General conditions: • 3o and 2o halides • strong bases
E2 Reactions • In the presence of a strong base, elimination generally occurs in a concerted reaction via an E2 mechanism
E2 Reactions • SN2 reactions require an unhindered methyl or 1o halide • steric hinderance prevents nucleophile from attacking 3o halides and forming the substitution product • E2 reactions generally involve the reaction between a 3o and 2o alkyl halides and a strong base.
E2 Reactions • The reaction of t-butyl bromide with methoxide ion gives only the elimination product. • The base attacks the alkyl bromide much faster than the bromide can ionize.
E2 Reactions • Many alkyl halides can eliminate in more than one way. • Mixture of alkenes produced
E2 Reactions • Saytzeff Rule: • When two or more elimination products can be formed,the product with the most highly substituted double bond will usually predominate. R2C=CR2 > R2C=CHR > RHC=CHR and R2C=CH2 > RHC=CH2
NaOCH2CH3 EtOH E2 Reactions Example: Draw the structures for all possible products of the following reaction. Which one will predominate?
E2 Reactions • E2 reactions follow a concerted mechanism: • bonds breaking and forming simultaneously • specific geometry required to allow overlap of orbitals of bonds being broken and bonds being formed • E2 reactions commonly involve an anti-coplanar conformation.
E2 Reactions Example: Predict the structure of the elimination product formed by the following reaction. NaOCH3 CH3OH
E1 vs E2 E2 E1 • Weak base • 30 > 2o • Good ionizing solvent • polar, protic (water, alcohols) • Saytzeff product • No required geometry • Rearranged products possible • Strong base required • 3o > 2o • Solvent polarity not important • Saytzeff product • Coplanar leaving groups (usually anti) • No rearrangements