S N 1 Reactions
200 likes | 277 Views
Learn about SN1 reactions, including reaction mechanism, key factors affecting the reaction, stability of carbocations, role of nucleophiles and leaving groups, solvent effects, stereochemistry, and potential rearrangements.

S N 1 Reactions
E N D
Presentation Transcript
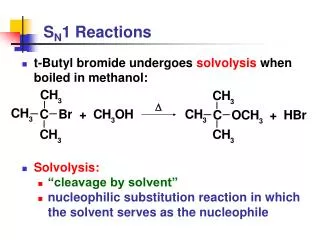
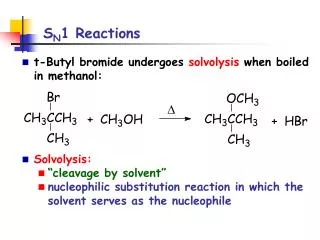
SN1 Reactions • SN1: substitution, nucleophilic, unimolecular rate = k[substrate] • Solvolysis is an SN1 reaction in which the solvent is nucleophile. (CH3)3CBr +CH3OH (CH3)3COCH3 +HBr boil t-butyl bromide Nuc methyl t-butyl ether
SN1 Reaction Mechanism • The substrate forms a carbocation intermediate. • The carbocation is planar with an empty 2p orbital available for bonding. 2p
SN1 Reaction Mechanism • The carbocation intermediate quickly reacts with the nucleophile. • Attack can be from either side. +
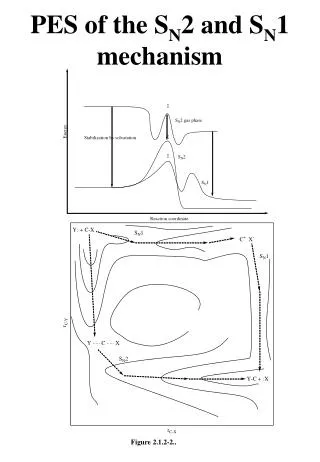
SN1 Reaction Profile rate = k[(CH3)3CBr] -EA(step 1)/RT k = Ae
SN1 Reactions • SN1 reactions are exothermic. • SN1 reactions occur in at least two steps. • The first step is slow.It is the formation of the carbocation intermediate, which is a very strong electrophile. • The second step is very fast. It is the reaction of the carbocation with the nucleophile.
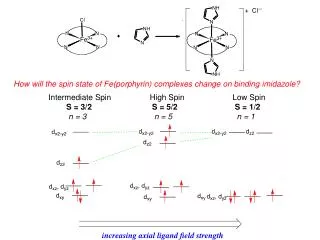
Factors Affecting SN1 Reactions • Structure of the substrate • Can a stable carbocation be formed? • Strength of the nucleophile • Nature of the leaving group • The solvent in which the reaction is run. • Must be able to stabilize the carbocation and the LG (which is usually an ion).
Factors Affecting SN1 Reactions - Structure of the Substrate • The carbocation is stabilized by any alkyl groups that are bonded to the electrophilic C atom. • inductive effect from σ bonds • hyperconjugation with p orbital • Resonance will also stabilize the carbocation. • Relative rates for SN1: 3°>2°>1°(resonance)>”1°”
Stability of the Carbocation - Hyperconjugation • Alkyl groups stabilize the positive charge through hyperconjugation, a partial overlap of an sp3 orbital with the empty 2p orbital of the C atom. • The more alkyl groups bonded to the electrophilic C, the more stabilization.
Stability of the Carbocation • This is the most important factor influencing the rate of SN1 reactions. • Stability: 3° > 2° >1° > methyl > > > methyl 3° 2° 1°
Carbocations for SN1 • SN1 reactions only occur for 3°, 2°, allylic, and benzylic cations. benzylic* 2° 3° allylic* *The H’s can be R’s.
Factors Affecting SN1 Reactions - Strength of the Nucleophile • The rate is not much affected by the nucleophile. It may be weak, moderate, or strong. • The carbocation is such a strong electrophile that any nucleophile will do.
Factors Affecting SN1 Reactions - the Leaving Group • The LG should be excellent. • Review: a good LG must be • electron withdrawing, to polarize the bond and make the C atom electrophilic, • polarizable, to stabilize the transition state, and • stable in the solvent (so it cannot be a strong base). Best LGs are neutral species or anions with a stabilized charge.
Factors Affecting SN1 Reactions - the Leaving Group • The leaving group (LG) serves two purposes in an SN1 reaction. These are the same as for the SN2 reaction. • It polarizes the bond that makes the C atom electrophilic. • It carries away a pair of electrons from the electrophilic C atom.
Factors Affecting SN1 Reactions - Solvent Effects • The solvent must be capable of dissolving both the carbocation and the leaving group. • SN1 reactions require highly polar solvents that strongly solvate ions. • Typical solvents: water, an alcohol, acetone (to help the alkyl halide to dissolve), or a mix.
Stereochemistry of the SN1 Reaction • Because the carbocation is planar, the nucleophilic attack can come from either side. -H+ -H+ • The products have more inversion, because the LG partially blocks that side.
Rearrangements in SN1 Reactions • The carbocation can undergo a structural rearrangement to produce a more stable species. • hydride shift (~H) • methyl shift (~CH3) • If ionization would lead to a 1° carbocation, look for a rearrangement to occur.
Mechanism of the Hydride Shift • In this case, the hydride shift occurs at the same time the Br leaves…why? CH3OH ~H -H+
Mechanism of the Methyl Shift • Sometimes the rearrangement occurs as a separate step…when? CH3OH ~CH3 -H+
SN1 Reactions - Summary • The structure of the carbocation is the most important factor: • Relative rates for SN1: 3°>2°>”1°”. • The nucleophile is typically weak or moderate in strength. • The LG should be excellent. • The solvent be polar and protic to stabilize the carbocation and LG. • Products will exhibit racemization and possibly rearrangements.