Understanding the Properties of Water: Polarity, Solubility, and pH
DESCRIPTION
This section explores the essential properties of water, including its polar nature and the significance of polar covalent bonds. Water is known as the universal solvent, capable of dissolving polar substances due to hydrogen bonding. We also discuss cohesion and adhesion, illustrated with examples like the water strider and the Jesus lizard. Additionally, we touch on temperature effects and the characteristics of solutions, acids, and bases, highlighting the importance of water in chemical reactions and life processes.
1 / 8
Download Presentation 

Understanding the Properties of Water: Polarity, Solubility, and pH
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
Properties of Water - Polarity Polar Covalent bonds
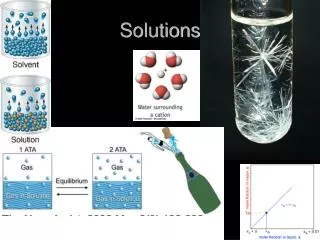
Solubility Water can dissolve polar substances Universal solvent
Cohesion and Adhesion water strider videojesus lizard video video
More Related

Audio
Live Player