BRAIN DEVELOPMENT AND PLASTICTY
BRAIN DEVELOPMENT AND PLASTICTY. Dr. Nelly Amalia Risan, SpA(K). Divisi Neuropediatri Departemen Ilmu Kesehatan Anak Fakultas Kedokteran Universitas Padjadjaran. Embryonic and Fetal Development of the Human Brain. Actual Size. Actual Size. Photographs of Human Fetal Brain Development.

BRAIN DEVELOPMENT AND PLASTICTY
E N D
Presentation Transcript
BRAIN DEVELOPMENT AND PLASTICTY Dr. Nelly Amalia Risan, SpA(K) Divisi Neuropediatri Departemen Ilmu Kesehatan Anak Fakultas Kedokteran Universitas Padjadjaran
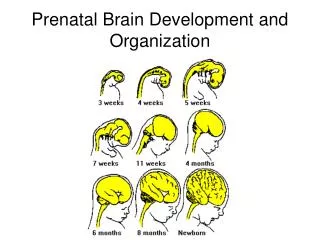
Embryonic and Fetal Development of the Human Brain Actual Size Actual Size
Photographs of Human Fetal Brain Development Lateral view of the human brain shown at one-third size at several stages of fetal development. Note the gradual emergence of gyri and sulci.
Nervous System Development in the Human Embryo (a) At 18 days after conception the embryo begins to implant in the uterine wall. It consists of 3 layers of cells: endoderm, mesoderm, and ectoderm. Thickening of the ectoderm leads to the development of the neural plate (inserts).(b) The neural groove begins to develop at 20 days.
Nervous System Development in the Human Embryo (c) At 22 days the neural groove closes along the length of the embryo making a tube. (d) A few days later 4 major divisions of the brain are observable – the telencephalon, diencephalon, mesencephalon, and rhombencephalon.
Neuroplasticity The brain consists of nerve cells (or "neurons") and glialcell which are interconnected, and learning may happen through change in the strength of the connections, by adding or removing connections, and by the formation of new cells.
"Plasticity" relates to learning by adding or removing connections, or adding cells.
Eight Phases in Embryonic and Fetal Development at a Cellular Level • Mitosis/Proliferation • Migration • Differentiation • Aggregation • Synaptogenesis • Neuron Death • Synapse Rearrangement • Myelination 8 stages are sequential for a given neuron, but all are occurring simultaneously throughout fetal development
Eight Phases in Embryonic and Fetal Development at a Cellular Level 1. Mitosis 2. Migration 3. Aggregation and 4. Differentiation 5. Synaptogenesis 6. Death 7. Rearrangement 8. Myelination
1. Mitosis/Proliferation • Occurs in ventricular zone • Rate can be 250,000/min • After mitosis “daughter” cells become fixed post mitotic
1. Mitosis/Proliferation: Neurons and Glia At early stages, a stem cell generates neuroblasts. Later, it undergoes a specific asymmetric division (the “switch point”) at which it changes from making neurons to making glia
2. Migration Note that differentiation is going on as neurons migrate.
2. Migration Radial Glia Radial glial cells act as guide wires for the migration of neurons
Growth cones crawl forward as they elaborate the axons training behind them. Their extension is controlled by cues in their outside environment that ultimately direct them toward their appropriate targets. 2. Migration Growth Cones The fine threadlike extensions shown in red and green are filopodia, which find adhesive surfaces and pull the growth cone and therefore the growing axon to the right.
2. Migration Growth Cones Scanning electron micrograph of a growth cone in culture. On a flat surface growth cones are very thin. They have numerous filopodia Ramon y Cajal drew these growth cones showing their variable morphology
2. Migration: How Do Neurons “Know” Where to Go? There are extrinsic and intrinsic determinants of neurons’ fate. • Extrinsic signals • Different sources of extrinsic signals • Generic signal transduction pathway • Intrinsic determinants
Migration During development, each of the three cells arise from different regions of the growing embryo. The Schwann cells arise from the neural crest and are led by the axons to their destination. Upon reaching it, they form a loose, unmyelinated covering over the innervating axons.
Migration The movement of the axons (and subsequently the Schwann cells) is guided by the growth cone, a filamentous projection of the axon that actively searches for neurotrophins released by the myotube
3. Differentiation • Neurons become fixed post mitotic and specialized • They develop processes (axons and dendrites) • They develop NT-making ability • They develop electrical conduction
3. Differentiation Development of the cerebral cortex The ventricular zone (VZ) contains progenitors of neurons and glia. 1st neurons establish the preplate (PP); their axons an ingrowing axons from the thalamus establish the intermediate zone (IZ). Later generated neurons establish layers II-VI. After migration and differentiation there are 6 cortical layers.
Differentiation At Pre-synaptic Differentiation, the changes exhibited at the developing axon terminal are well characterized. The pre-synaptic axon shows an increase in synaptic volume and area, an increase of synaptic vesicles, clustering of vesicles at the active zone, and polarization of the pre-synaptic membrane.
Differentiation The post-synaptic end plate grows deeper and creates folds through invagination to increase the surface area available for neurotransmitter reception.
Differentiation At birth, Schwann cells form loose, unmyelinated covers over groups of synapses, but as the synapse matures, Schwann cells become dedicated to a single synapse and form a myelinated cap over the entire neuromuscular junction
4. Aggregation Like neurons move together and form layers
5. Synaptogenesis Axons (with growth cones on end) form a synapse with other neurons or tissue (e.g. muscle)
Synaptogenesis Synaptogenesis is the formation of synapses. Although it occurs throughout a healthy person's lifespan, an explosion of synapse formation occurs during early brain development. Synaptogenesis is particularly important during an individual's "critical period" of life, during which there is a certain degree of neuronal pruning due to competition for neural growth factors by neurons and synapses.
Synaptic plasticity The underlying principle of synaptic plasticity is that synapses undergo and activity-dependent and selective strengthening or weakening so new information can be stored Synaptic plasticity depends on numerous factors including the threshold of the presynaptic stimulus in addition to the relative concentrations of neurotransmitter molecules.
5. Synaptogenesis: Attraction to Target Cells Target cells release a chemical that creates a gradient (dots) around them. Growth cones orient to and follow the gradient to the cells. The extensions visible in c are growing out of a sensory ganglion (left) toward their normal target tissue. The chemorepellent protein Slit (red) in an embryo of the fruit fly repels most axons.
6. Neuron Death • Between 40 and 75 percent of all neurons born in embryonic and fetal development do not survive. • They fail to make optimal synapses.
Neuron Death Leads to Synapse Rearrangement Release and uptake of neurotrophic factors Neurons receiving insufficient neurotropic factor die Axonal processes complete for limited neurotrophic factor
7. Synapse Rearrangement • Active synapses likely take up neurotrophic factor that maintains the synapse • Inactive synapses get too little trophic factor to remain stable
7. Synapse Rearrangement Time-lapse imaging of synapse elimination Two neuromuscular junctions (NM1 and NMJ2) were viewed in vivo on postnatal days 7, 8, and 9.
Spontaneous Network Activity During the early development of neural connections, excitatory synapses undergo spontaneous activation, resulting in elevated intracellular calcium levels which signals the onset of innumerable signaling cascades and developmental processes.
Teratogens Greek – “teratos” – wonder or monster “genos” - birth • Physical agents (e.g., x-rays) • Chemicals (e.g., drugs) • Microorganisms (e.g., rubella)