Scuba Diving and Its Chemistry
80 likes | 366 Views
Scuba Diving and Its Chemistry. SCUBA?. What does scuba stand for? Scuba stands for - S elf- C ontained U nderwater B reathing A pparatus. How does it work?

Scuba Diving and Its Chemistry
E N D
Presentation Transcript
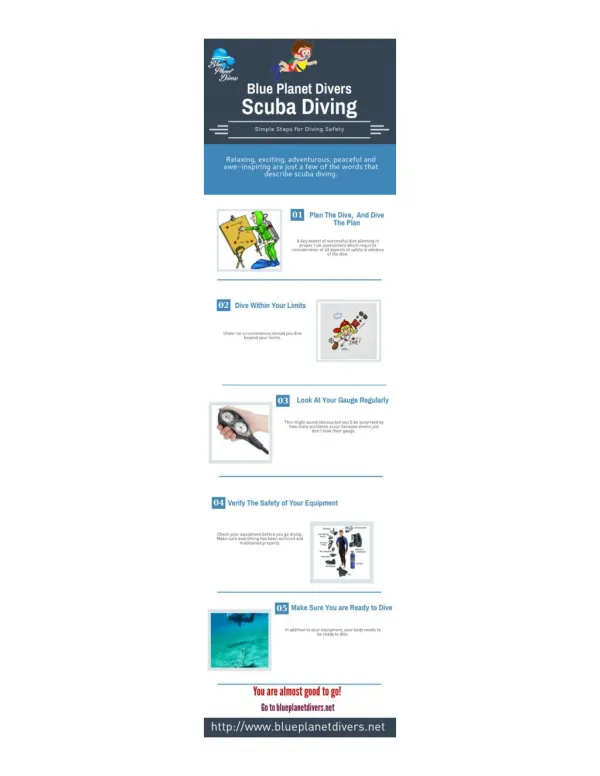
SCUBA? What does scuba stand for? Scuba stands for - Self-Contained Underwater Breathing Apparatus. How does it work? Scuba diving makes human mobile to see under water creatures and explore exotic places under the sea. Over the years Scuba technology has advanced, which means equipment is easier to use and and its, safer and more affordable.
Boyle’s Law? • Boyle's law describes the inversely proportional relationship between the absolute pressure and volume of a gas, if the temperature is kept constant within a closed system. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662. • Equation Below: P1 V1= 1 or P1 V1 = P2 V2P2 V2 Boyle's Law
Charles' Law? • At constant pressure, the volume of a given mass of an ideal gas increases or decreases by the same factor as its temperature (in Kelvin) increases or decreases. The law was published by Joseph Louis Gay-Lussac in 1802. • Equation Below: V = K T
Daltons' Law? • States that the total pressure exerted by a gaseous mixture is equal to the sum of the partial pressures of each individual component in a gas mixture. This empirical law was observed by John Dalton in 1801 and is related to the ideal gas laws. • Equation Below: PT = P1 + P2 + ..... + Pn
The Bends? • The bends can also be recognized as Decompression Sickness. Decompression sickness can be defined as a condition arising from the precipitation of dissolved gasses into bubbles inside the body on depressurization.
Air embolisms? • Air embolisms can be defined as tiny bubbles of air being transmitted into the bloodstream which could led to a defect in the human body.
Oxygen Toxicity? • Oxygen Toxicity can be defined as a condition resulting from the harmful effects of breathing molecular oxygen (O2) at elevated partial pressures.