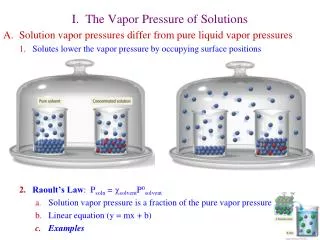
Vapor Pressure
Vapor Pressure. Number of Molecules. cool liquid. Temperature (speed). boiling point. Kinetic-Molecular Model. Thermometers only measure average speed. In any sample of a liquid there will be some faster, hotter molecules and some slower, colder ones.

Vapor Pressure
E N D
Presentation Transcript
Number of Molecules cool liquid Temperature (speed) boiling point Kinetic-Molecular Model Thermometers only measure average speed. In any sample of a liquid there will be some faster, hotter molecules and some slower, colder ones. The average temperature of molecules in this liquid is well below the boiling point. The small fraction of molecules with a speed above that of the b.p. will evaporate.
sealed bottle At the Molecular Level Most of the water molecules are down here, bound together as a liquid. They are shown as blue circles. A few water molecules are up here, as water vapor. They are mixed in with the rest of the gases in the air, shown as white circles. THE PRESSURE THESE EVAPORATED MOLECULES CONTRIBUTE TO THE AIR PRESSURE IN THE BOTTLE IS CALLED “VAPOR PRESSURE”. Individual molecules “pop” back and forth between states as they collide and gain or lose KE. But if the temperature is constant the total numbers of each remain constant.
warm liquid Number of Molecules cool liquid Temperature (speed) boiling point Kinetic-Molecular Model When the liquid warms up, its average speed increases. Now a larger portion of the molecules can evaporate.
sealed bottle At the Molecular Level As the water warms up, more evaporation occurs. H2O molecules crowd into the gas state as water vapor. The vapor pressure rises. A new balance is formed between the gaseous water vapor and the liquid water.
An Open Jar In an open jar balance is harder to reach. Water molecules which evaporate can leave the jar, allowing more water molecules to evaporate. In most situations, eventually the jar will lose all its water and contain only gases.
warm liquid Number of Molecules cool liquid boiling liquid Temperature (speed) boiling point Kinetic-Molecular Model When the average speed of the molecules equals the boiling point, the whole liquid boils. Evaporation can occur at any temperature, but boiling only occurs when the KE of the average liquid molecule is enough to let it change into the gas state.
Boiling Defined • Boiling occurs when the equilibrium vapor pressure above a liquid equals outside pressure.
Phase Diagrams: CO2 liquid solid gas °K -50 °F 0 °F 0 °C 100 °F