Novel (dimethoxy)benzothiazoles
150 likes | 406 Views
Novel (dimethoxy)benzothiazoles. Program goal to synthesise and develop novel benzothiazole compounds that are active in Phortress-resistant cell lines to identify a clinical candidate from this novel class of agent. Program status US Patent application filed 2004

Novel (dimethoxy)benzothiazoles
E N D
Presentation Transcript
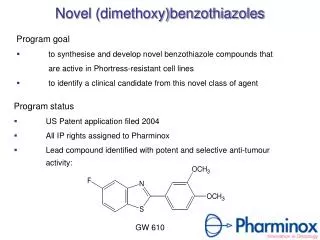
Novel (dimethoxy)benzothiazoles • Program goal • to synthesise and develop novel benzothiazole compounds that are active in Phortress-resistant cell lines • toidentify a clinical candidate from this novel class of agent Program status • US Patent application filed 2004 • All IP rights assigned to Pharminox • Lead compound identified with potent and selective anti-tumour activity: GW 610
Synthetic accessibility SAR: 5-F > 4-F > 6F > H; 3,4-dimethoxyphenyl preferred
In vitro antitumour activity: role of fluorine: R = 5F R = H Lung Colon Breast
Activity in MCF-7 and MDA 468 cells (CYP1A1 inducible) retained in presence of CYP1A1 inhibition by resveratrol. Active in HCC2998 and KM12 colon carcinoma cells - devoid of constitutive or inducible CYP1A1 Activity in cell lines with acquired resistance to DF 203: X 203R DF 203 √ X X 610R GW 610 GW 610 Activity- Independent of CYP 1A1 metabolism
Mechanism(s) of action • Independent of CYP 1A1 metabolism considerations - inactive analogues - CYP 1A1 non-inducible cell lines - inactivation of CYP 1A1 • Inhibits CYP 1A1 activity (IC50 8 μM) • Selective drug uptake in sensitive cells only • Selective generation of DNA adducts in sensitive cell lines only MCF-7, MDA 468, HCC 2998, KM12
GW 610 generates DNA adducts in sensitive cells only MDA 435 HCT 116 MCF-7 MDA 468 KM12 HCC 2998 1809.38 Adducts/108 Nucleotides 2628.62 Adducts/108 Nucleotides 677.90 Adducts/108 Nucleotides 1961.90 Adducts/108 Nucleotides
956.10 Adducts/108 Nucleotides 216.54 Adducts/108 Nucleotides DNA adduct generation associated with antitumour activity of 4F 610 6068.09 Adducts/108 Nucleotides MCF-7 Lung Breast 1627.65 Adducts/108 Nucleotides MDA 468 KM12 HCC 2998
Future work ●Identification of mechanism(s) of action ●Microarray / Proteomics studies ●Elucidation of structure / antitumour activity relationships ●Synthesis of pharmaceutically tractable analogues ●In vitro / in vivo profiling ●Selection of a clinical candidate
Novel (dimethoxy)benzothiazolesSummary and future outlook • Summary • a new series of synthetically accessible small molecules with potent and selective antitumour properties • mechanism of action distinct from structurally related 2-(4- aminophenyl)benzothiazoles (Phortress) Future Outlook • completion of SAR • optimisation of pharmaceutical properties and in vivo antitumour activity • scale-up chemistry • identification of first-in-class development candidate
A B MCF-7 (A) and HCC 2998 (B) cells were treated with DF 203 (NSC 674495) or compound 1 (NSC 721648) alone or in combination with resveratrol (5 μM; an inhibitor of CYP1A1 activity). In MCF-7 cells (CYP1A1-inducible) the activity of DF 203 was abolished. However, compound 1 still potently inhibited the growth of these cells. In HCC 2998 cells, possessing neither constitutive nor inducible CYP1A1, resveratrol failed to compromise the growth inhibitory properties of both agents.
Induction of cyp1a1 mRNA: 5F 203 and GW 610 5F 203, GW 610 sensitive □5F 203; NSC 703786 GW 610; NSC 721648 58 64 GW 610sensitive 5F 203, GW 610 Treatment 1 μM, 24 h
Inhibition of CYP1A1 activity by GW 610 The de-ethylation of ethoxyresorufin by CYP1A1 microsomes was measured in the absence and presence of increasing concentrations of GW 610. DF 203 was used as a positive control.














![Novel synthesis and antibacterial activity of 3-amino-8-chloro-4-oxo(2H)/Aryl/Heteryl-pyrazolo [3′,4′: 4,5] pyrimido](https://cdn4.slideserve.com/7842263/available-on-line-www-jocpr-com-dt.jpg)