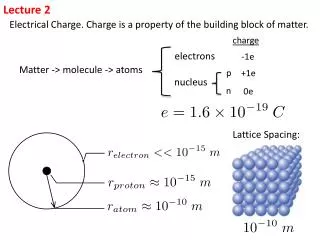
Lecture 2
A. Nitzan, Tel Aviv University ELECTRON TRANSFER AND TRANSMISSION IN MOLECULES AND MOLECULAR JUNCTIONS AEC, Grenoble, Sept 2005. Lecture 2. Grenoble Sept 2005. (1) Relaxation and reactions in condensed molecular systems Kinetic models Transition state theory

Lecture 2
E N D
Presentation Transcript
A. Nitzan, Tel Aviv University ELECTRON TRANSFER AND TRANSMISSION IN MOLECULES AND MOLECULAR JUNCTIONS AEC, Grenoble, Sept 2005 Lecture 2
Grenoble Sept 2005 • (1) Relaxation and reactions in condensed molecular systems • Kinetic models • Transition state theory • Kramers theory and its extensions • Low, high and intermediate friction regimes • Diffusion controlled reactions Coming March 2006 Chapter 13-15
Frequency dependent friction MARKOVIAN LIMIT WIDE BAND APPROXIMATION
Dielectric solvation Born solvation energy
Continuum dielectric theory of solvation WATER: tD=10 ps tL=125 fs
Electron solvation Quantum solvation (1) Increase in the kinetic energy (localization) – seems NOT to affect dynamics (2) Non-adiabatic solvation (several electronic states involved)
Activated rate processes Diffusion controlled rates KRAMERS THEORY: Low friction limit High friction limit Transition State theory
The physics of transition state rates Assume: (1) Equilibrium in the well (2) Every trajectory on the barrier that goes out makes it THIS IS AN UPPER BOUND ON THE ACTUAL RATE! Quantum barrier crossing:
PART B Electron transfer
Grenoble Sept 2005 • (2) Electron transfer processes • Simple models • Marcus theory • The reorganization energy • Adiabatic and non-adiabatic limits • Solvent controlled reactions • Bridge assisted electron transfer • Coherent and incoherent transfer • Electrode processes Coming March 2006 Chapter 16
Theory of Electron Transfer • Activation energy • Transition probability • Rate – Transition state theory Transition rate • Rate – Solvent controlled • NOTE: “solvent controlled” is the term used in this field for the Kramers low friction limit.
Electron transfer in polar media • Electron are much faster than nuclei • Electronic transitions take place in fixed nuclear configurations • Electronic energy needs to be conserved during the change in electronic charge density Electronic transition Nuclear relaxation
Electron transfer Nuclear motion Nuclear motion Electron transition takes place in unstable nuclear configurations obtained via thermal fluctuations
Electron transfer Solvent polarization coordinate
Transition state theory of electron transfer Alternatively – solvent control Adiabatic and non-adiabatic ET processes Landau-Zener problem (For diabatic surfaces (1/2)KR2)
Solvent controlled electron transfer Correlation between the fluorescence lifetime and the longitudinal dielectric relaxation time, of 6-N-(4-methylphenylamino-2-naphthalene-sulfon-N,N-dimethylamide) (TNSDMA) and 4-N,N-dimethylaminobenzonitrile (DMAB) in linear alcohol solvents. The fluorescence signal is used to monitor an electron transfer process that precedes it. The line is drawn with a slope of 1. (From E. M. Kosower and D. Huppert, Ann. Rev. Phys. Chem. 37, 127 (1986))
Electron transfer – Marcus theory We are interested in changes in solvent configuration that take place at constant solute charge distribution They have the following characteristics: (1) Pn fluctuates because of thermal motion of solvent nuclei. (2) Pe , as a fast variable, satisfies the equilibrium relationship (3) D= constant (depends on only) Note that the relations E = D-4P; P=Pn + Pe are always satisfied per definition, however D sE. (the latter equality holds only at equilibrium).
Electron transfer – Marcus theory Free energy associated with a nonequilibrium fluctuation of Pn q “reaction coordinate” that characterizes the nuclear polarization
The Marcus parabolas Use q as a reaction coordinate. It defines the state of the medium that will be in equilibrium with the charge distribution rq. Marcus calculated the free energy (as function of q) of the solvent when it reaches this state in the systems q =0 and q=1.
Electron transfer: Activation energy Reorganization energy Activation energy
Experimental confirmation of the inverted regime Marcus papers 1955-6 Miller et al, JACS(1984) Marcus Nobel Prize: 1992
Electron transfer – the coupling • From Quantum Chemical Calculations • The Mulliken-Hush formula • Bridge mediated electron transfer
Donor-to-Bridge/ Acceptor-to-bridge Bridge Green’s Function Franck-Condon-weighted DOS Reorganization energy Marcus expresions for non-adiabatic ET rates
Bridge mediated ET rate b’ (Å-1)= 0.2-0.6 for highly conjugated chains 0.9-1.2 for saturated hydrocarbons ~ 2 for vacuum
Bridge mediated ET rate Charge recombination lifetimes in the compounds shown in the inset in dioxane solvent. (J. M. Warman et al, Adv. Chem. Phys. Vol 106, 1999). The process starts with a photoinduced electron transfer – a charge separation process. The lifetimes shown are for the back electron transfer (charge recombination) process.
constant Incoherent hopping STEADY STATE SOLUTION
Dependence on temperature The integrated elastic (dotted line) and activated (dashed line) components of the transmission, and the total transmission probability (full line) displayed as function of inverse temperature. Parameters are as in Fig. 3.
The photosythetic reaction center Michel - Beyerle et al
Steady state evaluation of rates • Rate of water flow depends linearly on water height in the cylinder • Two ways to get the rate of water flowing out: • Measure h(t) and get the rate coefficient from k=(1/h)dh/dt • Keep h constant and measure the steady state outwards water flux J. Get the rate from k=J/h • = Steady state rate h