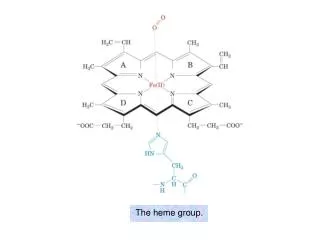
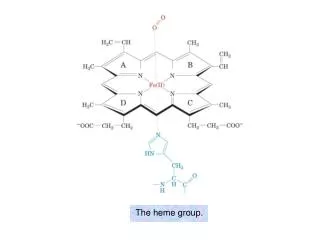
The heme group.
The heme group. The visible absorption spectra of oxygenated and deoxygenated hemoglobins. Oxygen dissociation curves of Mb and of Hb in whole blood. Hill plots for Mb and purified (“stripped”) Hb.

The heme group.
E N D
Presentation Transcript
The visible absorption spectra of oxygenated and deoxygenated hemoglobins.
A picket-fence Fe(II)–porphyrin complex with bound O2 (prevents auto-oxidation via dimerization)
Effect of pH on the O2-dissociation curve of Hb: the Bohr effect. Hb(O2)nHx + O2 Hb(O2)n+1 + xH+ x ≈ 0.6
CO2 + H2OH+ + HCO3- catalyzed by carbonic anhydrase in erythrocytes Carbamate Formation (N-termini) R-NH2 + CO2R-NH-COO- + H+ DeoxyHb binds more CO2 as carbamate than does oxyHb
The effect of 2,3-BPG on Hb oxygen affinity Comparison of the O2-dissociation curves of “stripped” Hb and whole blood in 0.01M NaCl at pH 7.0.
The effects of 2,3-BPG and CO2, both separately and combined, on hemoglobin’s O2-dissociation curve compared with that of whole blood (red curve).
The effect of high-altitude exposure on the p50 and the BPG concentration of blood in sea level–adapted individuals.
The O2-dissociation curves of blood adapted to sea level (black curve) and to high altitude (red curve).
Contains 8 helices: A-H Contains some 310 helices Subunits of Hb are similar to Mb Structure of sperm whale myoglobin (Mb)
The Amino Acid Sequences of the a and b Chains of Human Hemoglobin and of Human Myoglobin
The Amino Acid Sequences of the a and b Chains of Human Hemoglobin and of Human Myoglobin
Heme located in a hydrophobic pocket formed mainly by helices E and F Fe(II) is 0.22 Å out of the heme plane in oxyMb on the proximal His side; O2 in bent geometry Fe(II) is 0.55 Å out of plane in deoxyMb Structures of oxyMb and deoxyMb are superimposable Stereo drawings of the heme complex in oxyMb.
Contains two ab protomers Tertiary structures of a and b subunits are similar to each other and to Mb There is extensive interactions between unlike subunits (a1-b1 and a2-b2); hydrophobic in character Contacts between like subunits few and polar The X-ray structure of deoxyHb as viewed down its exact 2-fold axes.
Extensive quaternary structural changes occur to Hb upon oxgenation Changes occur at the a1-b2 and a2-b1 interfaces The X-ray structure of oxyHb as viewed down its exact 2-fold axes.
Oxygenation rotates thea1-b1 dimer by 15o with respect to the a2-b2dimer; two-fold symmetry is maintained 4o forms: deoxyHb = Tstate (tense) oxyHb = Rstate (relaxed) The major structural differences between the quaternary conformations of (a) deoxyHb and (b) oxyHb
Explaining cooperativity: Perutz mechanism (based on X-ray analyses) Note out-of-plane Fe(II) in deoxyHb; ion moves in-plane in oxyHb, and pulls on the proximal His; F helix is moved The heme group and its environment in the unliganded a chain of human Hb.
Triggering mechanism for the T ® R transition in Hb (T = blue; R = pink)
No stable intermediate states are allowed: a binary switch The a1C–b2FG interface of Hb in (a) the T state and (b) the R state.
black: deoxyHb blue: oxyHb The hemoglobin a1b2 interface as viewed perpendicularly to Fig. 10-13.
Salt bridges must break in T to R transition Val-1 on a2: Bohr effect Networks of salt bridges and hydrogen bonds in deoxyHb. (a) Last two residues of the a chains.
His-146 on b2: Bohr effect Networks of salt bridges and hydrogen bonds in deoxyHb. (b) Last two residues of the b chains.
Relative free energies of the T and R states vary with fractional saturation Overall binding curve for Hb is a composite of the hyperbolic binding curves for pure T and R Free energy and saturation curves for O2 binding to hemoglobin
Hb with carbamoylated a subunits (N-terminal amino groups) lacks 20-30% of the Bohr effect. Reaction of cyanate with the unprotonated (nucleophilic) forms of primary amino groups.
BPG binding pocket is lined with positive charge (Lys, His, N-termini): complementary to BPG’s negative charge BPG preferentially binds to deoxyHb: central cavity is smaller in oxyHb Binding of BPG to deoxyHb: selective stabilization of the T form
Abnormal Hemoglobins: Hemoglobinopathies - 860 variant Hbs in humans Mutations stabilizing the Fe(III) oxidation state of heme. (a) Alterations in the heme pocket of the a subunit on changing from deoxyHbA to Hb Boston.
Mutations stabilizing the Fe(III) oxidation state of heme. (b) The structure of the heme pocket of the b subunit in Hb Milwaukee.
Sickle-Cell Anemia: HbS Single-site mutation: Valine replaces Glu A3(6)b Electron micrograph of deoxyHbS fibers spilling out of a ruptured erythrocyte.
220-Å in diameter fibers of deoxyHbS: an electron micrograph of a negatively stained fiber
220-Å in diameter fibers of deoxyHbS: a model, viewed in cross section, of the HbS fiber.
Structure of the deoxyHbS fiber: arrangement of the deoxyHbS molecules in the fiber.
Intermolecular association Val 6 involving b2; Val 6 of b1 - pocket Structure of the deoxyHbS fiber: a schematic diagram indicating the intermolecular contacts in the crystal structure of deoxyHbS.
Molecular Basis for Fibril Formation In HbS Structure of the deoxyHbS fiber: the mutant Val 6b2 fits neatly into a hydrophobic pocket formed mainly by Phe 85 and Leu 88 of an adjacent b1 subunit.
Note delay, td 1/td = k(ct/cs)n: concentration dependence of the delay time Time course of deoxyHbS gelation: the extent of gelation as monitored calorimetrically (yellow) and optically (purple).
Implies a 30th power concentration dependence Time course of deoxyHbS gelation: a log–log plot showing the concentration dependence of 1/tdfor the gelation of deoxyHbS at 30°C.
Allosteric regulation: two general models Monod, Wyman, Changeux: symmetry model conformational change alters affinity for ligand: molecular symmetry conserved The species and reactions permitted under the symmetry model of allosterism
Koshland, Nemethy, Filmer Binding to T-state induces conformational changes in unliganded subunits (intermediate affinity between T and R) The sequential model of allosterism
Ligand affinity varies with number of bound ligands; intermediate conformations Sequential binding of ligand in the sequential model of allosterism
The sequential and the symmetry models of allosterism can provide equally good fits to the measured O2-dissociation curve of Hb.
More complex model of Hb allosterism Free energy penalties for binding O2 to various ligation states of Hb tetramers relative to O2-binding to noncooperative Hb ab dimers.