Understanding Ionic and Molecular Compounds: Key Characteristics and Naming Conventions
This guide explores the properties and classification of ionic and molecular compounds. Ionic compounds form between metals and non-metals and exhibit distinct characteristics, such as high electronegativity differences and the ability to conduct electricity in aqueous solutions. The naming conventions for acids and bases are also outlined, including classical and IUPAC standards. In addition, the states of matter for various compounds at Standard Ambient Temperature and Pressure (SATP) are discussed, providing a comprehensive overview for students of chemistry.

Understanding Ionic and Molecular Compounds: Key Characteristics and Naming Conventions
E N D
Presentation Transcript
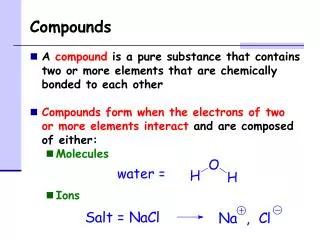
Ionic compounds • Compound between Non-metal and metal • Ionic bond • Large difference in electronegativity • (how much the atom wants an electron) • Found in solid form at SATP • Conducts electricity in aqueous solution • When combined the cation is used followed by the anion, the cation is used in full and the anion has a –ide suffix: oxide, fluoride
Cont’d • Multi valent metals • Different possibilities for ion charge • Iron, Copper • Check period table for common ion charge
Molecular Elements • Elements ending in –gen are diatomic. Pg. 33 – should know this table
Molecular Compounds • Compounds of non-metals • Covalent bond • Small difference in electronegativity • Found in all three states of matter at SATP • Does not conduce electricity
Acid and Base Compounds • Identified by the H- prefix and occasionally the –COOH on the end of the compound • Found in all three states of matter at SATP • Conduce electricity in aqueous solution • Made of non-metals – assumed to be ionic • Acid pH below 7 – aqueous hydrogen • Base pH above 7 – aqueous hydroxides
Acid naming • Classical naming (for information) • hydrogen _____ide -> hydro_____ic acid • hydrogen _____ate -> ______ic acid • hydrogen _____ite -> ________ous acid • hydrogen hypo_____-> hypo_____ous • IPUAC naming(good to know basic) • hydrogen ______
States of matter in a compound • The state of matter is also given in the compound • i.e. H20(s), H20(l), H20(g) • NaCl(s) or NaCl(aq) • Aqueous solution – the solvent is water (dissolved in water) - homogenous mixture in one phase • Solvent – a substance that dissolves a solute • Solute – substance that is dissolved into a solution