Thyroid and Anti-Thyroid Drugs
690 likes | 4.27k Views
Thyroid and Anti-Thyroid Drugs. An overview of the thyroid, thyroid drugs, and the mechanisms through which they affect thyroid function. The Thyroid. Thyroid hormones are synthesised by iodination of tyrosine residues on thyroglobulin within the lumen of the thyroid follicle.

Thyroid and Anti-Thyroid Drugs
E N D
Presentation Transcript
An overview of the thyroid, thyroid drugs, and the mechanisms through which they affect thyroid function.
The Thyroid Thyroid hormones are synthesised by iodination of tyrosine residues on thyroglobulin within the lumen of the thyroid follicle. The thyroglobulin is endocytosed and thyroxine (T4) and triiodothyronine (T3) are secreted. Synthesis and secretion of T3 and T4 are regulated by thyroid-stimulating hormone (TSH; thyrotrophin) and influenced by plasma iodide
T3 and T4 actions are: Stimulate metabolism generally, causing increased oxygen consumption and increased metabolic rate . Influence growth and development. Within cells, the T4 is converted to T3, which interacts with a nuclear receptor; the receptor represses basal transcription when not bound to T3, and activates transcription when bound.
There is a large pool of T4 in the body; it has a low turnover rate and is found mainly in the circulation. There is a small pool of T3 in the body; it has a fast turnover rate and is found mainly intracellular.
Abnormalities of thyroid function include: hyperthyroidism (thyrotoxicosis), either diffuse toxic goiter or toxic nodular goiter hypothyroidism; in adults this causes myxoedema, in infants, cretinism simple non-toxic goiter, caused by dietary iodine deficiency, usually with normal thyroid function
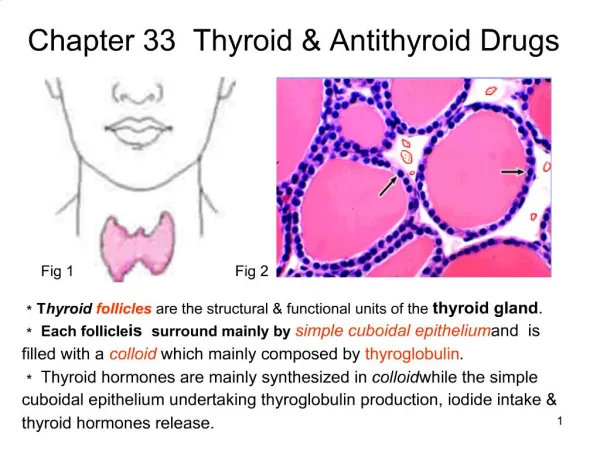
Anatomy and Physiology of the Thyroid Gland • Member of the Endocrine System • Secretes thyroid hormones, thyroxine and calcitonin, which regulate metabolism and growth. • Located in neck adjacent to the 5th cervical vertebra (C5). • Composed of epithelial cells which specialize in the absorption of iodine and, of course, secretion of thyroid hormones. • Follicles surround a protein core, the colloid, where thyroglobulin, a substrate in thyroid hormone synthesis, and thyroid hormones are stored.
Synthesis of Thyroid Hormones Regulation: • The hypothalamus in the brain secretes thyroid releasing hormone, TRH, that target the pituitary gland which, in turn, secretes thyroid stimulating hormone, TSH. The pituitary gland’s sensitivity toward TRH varies with the body’s need for thyroid hormones.
TSH is absorbed into the thyroid, stimulating the thyroid to absorb iodine and synthesize hormones. • Thyroid hormones provide negative feedback for TSH production via a “homeostatic feedback loop.”
TH Synthesis: • Thyroid peroxidase (TPO) catalyzes the conversion of iodide (I2) to iodine (I-) using H2O2 as a cofactor. • TPO then catalyzes the addition of iodine to the C-3 and C-5 position of a tyrosine residue of thyroglobulin. • Two iodinated thyrosine rings condense to form thyroxine, or T4, with four iodine substituents. • Triiodothyronine, or T3, with three iodine substituents, accounts for about 10% of thyroid hormone production.
TSH TSH acts on receptors on the membrane of thyroid follicle cells and its main second messenger is cAMP. It controls all aspects of thyroid hormone synthesis: the uptake of iodide by follicle cells, by stimulating transcription of the Na+/I- transporter gene; this is the main mechanism by which it regulates thyroid function .
the synthesis and secretion of thyroglobulin the generation of H2O2 and the iodination of tyrosine endocytosis and proteolysis of thyroglobulin secretion of T3 and T4 the blood flow through the gland. TSH also has a trophic action on the thyroid cells; it stimulates the action of the genes for thyroglobulin, thyroperoxidase and the Na+/I- transporter
Mechanism of TH Activation in Body Hydrophobic molecule transported in the bloodstream with a requisite carrier protein, TBG. Albumin also serves as a TH carrier protein. Transported across the cell membrane using a transporter complex. TH enters nucleus. The iodine at position 5 on the outer ring serves to sterically hinder the thyroid hormone binding enzyme. T4 is converted to T3, the active form. Deiodinase, specifically IDI or IDII, cleaves the iodine at position 5 to yield triiodithronine, T3.
TH Effects on Metabolism TH serves as a nuclear transcription factor, regulating gene expression in targeted cells to increase metabolism. Increase size and number of mitochondria in the cell. Synthesizes cytochromes which feed into the electron transfer chain of cellular respiration, stimulating metabolism through increasing ATP production. Increase ATPase concentration, the enzyme which cleaves a phosphate group from ATP forming ADP and inorganic phosphate. Increased K+ and Na+ concentrations in the cell.
Increase the body’s basal metabolic rate, BMR, to maintain electrochemical gradient in cell. Stimulate carbohydrate metabolism and lipolysis, or the break down of fats. Affects protein synthesis. Increase the body’s sensitivity to cathecholamines, i.e. adrenaline
Conditions that Impair Thyroid Function: Hypothyroidism Insufficient amount of thyroid hormone synthesized causing lethargy and weight gain, among other symptoms. Primary hypothyroidism is typically caused by Hashimoto’s Disease, an auto-immune disorder in which the thyroid is destroyed by antibodies. Impaired hypothalamus and pituitary function, typically due to a tumor, can inhibit the secretion of THS, causing secondary hypothyroidism. A diet insufficient in iodine causes hypothyroidism as well.
Symptoms of Thyroid Dysfunction: Goiter Enlarged thyroid, symptom of hypothyroidism. Goiters form for different reasons depending on the cause of hypothyroidism Hashimoto’s disease, also known as chronic lymphocytic thyroiditis, causes goiters due to the accumulation of lymphocytes. The decreased amount of thyroid hormones in the body, due to Hashimoto’s or other thyroid disorders including infection, signals the increased production of TSH which accumulates in the thyroid causing a characteristic goiter. Goiters form due to an insufficient amount of ingested iodine and serve to increase the surface area of the thyroid and aid in its absorption of iodine.
Thyroid hormones and iodine Thyroxine is the standard replacement therapy for hypothyroidism. Thyroxine has all the actions of endogenous thyroxine; it is given orally. Liothyronine (T3) is the treatment of choice for myxoedema coma. Liothyronine has all the actions of endogenous triiodothyronine; it is given intravenously
Treatment for Hypothyroidism Hormone replacement therapy Administered orally with a bioavailability ranging from 48%-80%. Levothyroxine—Synthetic T4 Liothyronine—Synthetic T3 Liotrix—Combination of synthetic T4 and T3 Natural Thyroid Hormones—Thyroid hormones derived from pigs, contains T4 and T3 Armour Thyroid .
Dosage specific to individual and is determined by their TSH serum levels. Typically 1.5μg T4 per kg body weight. Because thyroid hormones serve to increase heart rate, T4, the inactive form, is typically administered to older patients who have an increased risk for heart attack on account of their age. Synthetic T3 is reserved for younger patients, who do not have a history of heart problems and individuals non-responsive to T4 treatment.
Some men are inefficient in the conversion of T4 to T3, making combination drugs like Litrix and Armour Thyroid ideal treatment options. • Dosage for individuals suffering from secondary hypothyroidism determined by the amount of free T4 and T3 circulating in their system. • Administering too high of a dosage leads to hyperthyroid symptoms
Conditions that Impair Thyroid Function: Hyperthyroidism The over production of thyroid hormones. Symptoms include fatigue, weight lose, rapid heart beat, anxiety, swollen eyes, and sensitivity to hot temperatures.
Causes: Grave’s disease, and autoimmune disorder in which antibodies serve as agonists to the THS receptors on the thyroid’s surface, causing thyroid growth and activation of hormone synthesis and secretion. Thyroid tumors which cause the uncontrolled synthesis and secretion of thyroid hormones. Thyroiditis, inflammation of the thyroid typically caused by infection.
Treatment for Hyperthyroidism Anti-thyroid drugs—Thioureylenes Inhibits thyroid hormone synthesis by irreversibly binding to TPO inhibiting its ability to break down iodine (I2→I-) and covalently attach it to the tyrosine residue of thyroglobulin. Propylthiouracil Methimazole Carbamizole─Degraded to methimazole in the body. Thyroidectomy β-Blockers used in the treatment of thyroiditis to treat symptoms.
Carbimazole (or propylthiouracil) Hyperthyroidism (diffuse toxic goitre), at least 1 year of treatment being necessary; recurrence occurs eventually in over half the patients but can be managed by a repeat course of treatment. Propylthiouracil can be used in patients who suffer sensitivity reactions to carbimazole. Preliminary to surgery for toxic goitre. Part of the treatment of thyroid storm (very severe hyperthyroidism); propylthiouracil is preferred because of its action in decreasing the conversion of T4 to T3 in the tissues
Radioactive Iodine As first-line treatment for hyperthyroidism; recurrence is rare provided the dose is adequate. For treatment of relapse of hyperthyroidism after carbimazole therapy or surgery Radioiodine, given orally, is selectively taken up by thyroid and damages cells; it emits short-range β radiation, which affects only thyroid follicle cells. Hypothyroidism will eventually occur.
Iodine dissolved in aqueous potassium iodide ('Lugol's iodine') is used short-term to control thyrotoxicosis preoperatively. It reduces the vascularity of the gland. Iodine, given orally in high doses, transiently reduces thyroid hormone secretion and decreases vascularity of the gland.
Anti-Thyroid Drugs Effective in the long-term treatment of hyperthyroidism. 6-8 weeks before maximum effect of the drug achieved. Drug inhibits hormone synthesis, so hormones synthesized prior to drug use will continue to cause hyperthyroid condition. Typical side effects include headache, nausea, vomiting, itchy skin and rash, and muscle aches and pains. Serious liver damage, decreased red and white blood cell synthesis, as well as decreased platelet production have been reported in a few cases. The drug’s interaction with other enzymes responsible for clotting factor synthesis accounts for some of these serious side effects. Administering too high a dosage of anti-thyroid drugs can cause hypothyroidism.
Thyroid Treatment: Potential Drug Interactions Drugs that reduce thyroid hormone production Lithium Iodine-containing medications Amiodarone (Cordarone) Drugs that reduce thyroid hormone absorption Sucralfate (Carafate) Ferrous sulfate (Slow Fe) Cholestyramine (Questran) Colestipol (Colestid) Aluminum-containing antacids Calcium products
Drugs that increase metabolism of thyroxine Rifampin (Rifadin) Phenobarbital Carbamazepine (Tegretol) Warfarin (Coumadin) Oral hypoglycemic agents Drugs that displace thyroid hormone from protein binding Furosemide (Lasix) Mefenamic acid (Ponstel) Salicylates
TSH Replacement Drugs Thyrotropin alpha—A synthetic form of TSH. Administered intravenously. Used in thyroid cancer treatment. Tumors of the hypothalamus or pituitary gland can cause the uncontrolled release of TSH, which accumulates in the thyroid and can cause subsequent follicular or papillary cancer of the thyroid. Partial or total thyroidectomy typical. Following thyroidectomy, the individual is dependent on exogenous thyroid hormones to regulate metabolism, but thyrotropin alpha is also used to suppress the release of endogenous TSH, which could trigger cancerous growth again. Used as a diagnostic tool to determine the reoccurrence of cancer.
References http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=endocrin.section.280 http://www.rxlist.com/cgi/generic2/thyrotropin_cp.htm http://www.thyroid-info.com/drugs/armour.htm http://thyroid.about.com/cs/drugdatabase/f/thyrogen.htm http://www.parathyroid.com/hypoparathyroidism.htm http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=2482800&dopt=Abstract http://www.5mcc.com/Assets/SUMMARY/TP0466.html http://www.dundee.ac.uk/medther/tayendoweb/images/carbimazole.htm http://www.home.duq.edu/~harrold/Chem3D/thyroid_SAR_page_5.html http://www.aafp.org/afp/20011115/1717.html