Unit 3 Part 1 Atomic Structure
Unit 3 Part 1 Atomic Structure. ICP Mr. Patel SWHS. Topic Outline. Learn Major Elements The Atom Subatomic Particles Modern Atomic Theory Organizing the Elements Periodic Table Classifying Elements Atomic Models. Defining the Atom.

Unit 3 Part 1 Atomic Structure
E N D
Presentation Transcript
Unit 3 Part 1Atomic Structure ICP Mr. Patel SWHS
Topic Outline • Learn Major Elements • The Atom • Subatomic Particles • Modern Atomic Theory • Organizing the Elements • Periodic Table • Classifying Elements • Atomic Models
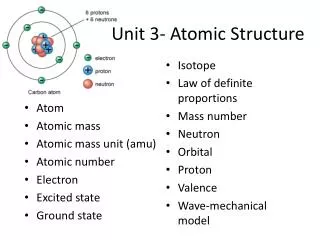
Defining the Atom • Atom – the smallest particle of an element that retains its identity • Can not see with naked eye • Nanoscale (10-9 m) • Seen with scanning tunneling electronmicroscope
Democritus • Democritus was a Greek to first come up with idea of an atom. • His belief: atoms were indivisible and indestructible. = WRONG! • Atom comes from “atmos” - indivisible
Dalton’s Atomic Theory • 2000 yrs later, John Dalton used scientific method to transform Democritus’s idea into a scientific theory • Dalton put his conclusions together into his Atomic Theory (4 parts)
Dalton’s Atomic Theory • All elements are composed of indivisible atoms.
Dalton’s Atomic Theory • Atoms of the same element are identical. Atoms of different elements are different
Dalton’s Atomic Theory • Atoms of different elements can chemically combine in whole number ratios.
Dalton’s Atomic Theory • Atoms of one element can never be changed to another element
The Electron • Particle with negative charge • Discovered by J.J. Thomson • Used cathode ray (electron) beam and a magnet/charged plate. • Millikan found the charge and mass
The Proton and Neutron • An atom is electrically neutral • If there is a negative particle then there must be positive particle • Proton – particle with positive charge • Chadwick discovered neutron – neutral charge
Thomson’s Atomic Model • Electrons distributed in a sea of positive charge • Plum Pudding Model
Rutherford’s Atomic Model • The nucleusis the central part of the atom containing protons and neutrons • Positive charge • Most of the mass • Electrons are located outside the nucleus • Negative charge • Most of the volume
Atomic Number • An element is defined only by the number of protons it contains • Atomic Number – number of protons • Number of protons = number of electron • For a neutral element
Identify the number of Protons • Zinc (Zn) • Iron (Fe) • Carbon (C) • Uranium (U) • 30 • 26 • 6 • 92
Mass Number • Nucleus contains most of the mass • Mass Number – total protons and neutrons Number of neutron = Mass # – Atomic #
Identify # of Subatomic Particles • Lithium (MN = 7) • Nitrogen(MN = 14) • Fluorine(MN = 19)**MN = Mass Number • 3 p+ , 3 e-, 4 n0 • 7 p+ , 7 e-, 7 n0 • 9 p+ , 9 e-, 10 n0
Differences in Particle Number • Different element: different number of protons • Ions – same number of proton, different number of electrons • Isotope – same number of proton, different number of neutrons • Different Mass Numbers
Two Notations for Atoms • Nuclear Notation • Write the element symbol • On left side, superscript = Mass Number • On left side, subscript = Atomic Number • Isotope –Hyphen Notation • Write full name of element • On right side, put a dash • On right side put Mass Number after dash Hydrogen - 3
Ex: Three isotopes of oxygen are oxygen-16, oxygen-17, and oxygen-18. Write the nuclear symbol for each.
Ex: Three isotopes of chromium are chromium-50, chromium-52, and chromium-53. How many neutrons are in each isotope?
Atomic Mass • Atomic Mass Unit (amu) – one-twelfth of the mass of the carbon-12 atom • Different isotopes have different amu (mass) and abundance (percentage of total) • Atomic Mass – weighted average mass of the naturally occurring atoms. • Isotope Mass • Isotope Abundance
Atomic Mass • Because abundance is considered, the most abundant isotope is typically the one with a mass number closest to the atomic mass. • Example, Boron occurs as Boron-10 and Boron-11. Periodic Table tells us Born has atomic mass of 10.81 amu. • Boron-11 must be more abundant