Unit 3 - Atomic Structure
Unit 3 - Atomic Structure. Chapter 5 Addison/Wesley Textbook. Earliest Model. 450 BC – Democritis, a Greek philosopher, first uses the word “atomos” which means indivisible Definition of atom today – Smallest particle of an element that still retains properties of that element. Late 1700’s.

Unit 3 - Atomic Structure
E N D
Presentation Transcript
Unit 3 - Atomic Structure Chapter 5 Addison/Wesley Textbook
Earliest Model • 450 BC – Democritis, a Greek philosopher, first uses the word “atomos” which means indivisible • Definition of atom today – Smallest particle of an element that still retains properties of that element
Late 1700’s • Lavoisier – Law of Conservation of Matter • Proust – Law of Constant Composition This says that the same compound from any source always contains the same elements in the same proportion by mass
First Atomic Theory • John Dalton • Proposed in 1803 • Compilation of other people’s work and a little of his own • Still true except for one part • Good biography: http://www.slcc.edu/schools/hum_sci/physics/whatis/biography/dalton.html
Dalton’s Atomic Theory • Each element is composed of tiny, indivisible atoms • Each element’s atoms are the same and unique • Atoms are only rearranged in any chemical reaction • A compound has the same number and kind of atom.
The Atom Today • Since 1981 we have “seen” atoms with a scanning tunneling microscope. • Uses a fine tip and a stable environment to trace the electronic field and image it on a computer • Lots of galleries on the Web: • http://physics.nist.gov/GenInt/STM/fig1.html
Discovery Atomic Structure • Early research comes from physicists’ work on electricity • “Electricity” is property of “electron”, which is amber • In ancient Greece, pieces of amber were rubbed and static electricity discharged • Ben Franklin did early research in late 1700’s
His work • Discovered two kinds of charges, positive and negative • Opposite charges attract • Like charges repel • Objects pick up charges • They discharge when touched to ground
Lightning • New research all the time • Great photography – Check YouTube • http://www.pbs.org/wgbh/nova/sciencenow/3214/02.html • http://www.teachersdomain.org/resource/phy03.sci.phys.mfw.lightning/
Electricity Research after Franklin • Physicists liked to zap things in the mid 1800’s • Cathode ray tube was device used by many (diagram) • Same device used as TV screen
How it Works • Metal is electrified in an evacuated tube • All metals gave a greenish ray going to the positive electrode • Ray could be attracted by a positive charge, repelled by a negative charge. • It could actually make a paddle wheel move - particle
Discovery of the Electron • JJ Thomson – Cavendish Lab - 1896 • Used cathode ray tube to determine amount of deflection • Determined that particle has a negative charge • Determined the charge to mass ratio of the particle • Animation: http://highered.mcgraw-hill.com/sites/0072512644/student_view0/chapter2/animations_center.html#
Finding the Charge of an Electron • American physicist – Robert Millikan • Famous Oil Drop Experiment (handout) • See animation
Explanation • Drops of oil are sprayed into a chamber • X-rays cause electrons to be formed • they cling to oil (in varying numbers) • Drops pass through a set of electric plates which have a charge • Millikan adjusted charge to balance the charge on each drop • Found the greatest common factor
Conclusion • Charge on an electron is 1.60 X 10-19 Coulombs • Mass of an electron is 9.11 X 10-19 grams • Virtually without mass
Discovery of Radiation • Henri Becquerel accidentally discovered radiation in 1896 • Photographic plate wrapped and put in drawer for weekend gets exposed • Rock was “radiating” something • Rock was pitchblende which contains radium
Characteristics of radiation • Spontaneously emitted by some elements • Studied by Marie and Pierre Curie • They discovered several elements, including uranium and polonium • Atom emits radiation and then changes • This gave clues to what atom is actually made of
Marie and Pierre Curie • Good site • http://www.aip.org/history/curie/
Further Research on Radiation • Ernst Rutherford is brought to Cavendish Lab in early 1900’s • Studied radioactivity • Analyzed nature of radiation • Handout
Magic Bullet • Alpha Particle chosen • Right size • Could be detected afterwards • Helium nucleus – 2 protons and 2 neutrons • +2 charge
Gold Foil Experiment • Rutherford got grad students to design set up • Geiger and Marsden • Wanted to confirm Thomson’s “Plum Pudding” model of the atom – electrons stuck in positive pudding • Handout
Explanation • Find a source of alpha particles • Aim them at a piece of gold foil • Check to see where they come out by counting fluorescent spots
Results • Most went through • Very small number were deflected almost straight back • Only explanation was that all matter was concentrated into a dense nucleus • Nucleus had a positive charge • Electrons traveled in empty space around the nucleus • Movie: Empty Space
Results • Most went through • Very small number were deflected almost straight back • Only explanation was that all matter was concentrated into a dense nucleus • Nucleus had a positive charge • Electrons traveled in empty space around the nucleus • Movie: Empty Space – next slide
Atom is Empty Space • From NOVA
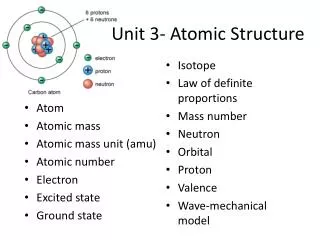
Modern Atomic Theory There are 3 major subatomic particles (protons, neutrons, and electrons). There are basic particles that make these up but we will not discuss them The proton also came from the cathode ray tube The neutron was discovered by Chadwick, a student of Rutherford in 1935.
Summary of Particles PROTON NEUTRON ELECTRON +1 NONE -1 LARGE LARGE VERY SMALL NUCLEUS NUCLEUS OUTSIDE NUCLEUS
Planetary Model • Proposed by Rutherford • Electrons orbit nucleus like planets around sun • Atoms are neutral so #protons = #electrons • Charge on electron: 1.602 X 10-19 C or “1” • Mass of proton: 1.67 X 10-24 g or 1 amu (atomic mass unit)
Atomic Number • Defined by Henry Mosely (1887-1915) • Student of Rutherford • Unique for each element • Number of protons in the nucleus • What is atomic number of nitrogen? Uranium?
Isotopes • Means “type or form” • All atoms of the same element have the same number of protons • There may be different types of the same elements, called isotopes • Vary in number of neutrons, mass • Try Carbon-12 and Carbon-14
Characteristics of Isotopes • Varying masses • Same chemical and physical properties • Some may be unstable, and therefore radioactive
Symbol • Carbon-12 • 12 is mass number, # protons + # neutrons • Also written 126C • Mass # - Atomic # = # of neutrons
Atomic Mass • Mass of an isotope in amu’s is simply the Mass number • Most elements have several common isotopes • Mass on periodic table must reflect this, that is why there are decimals • Weighted average calculation (like grades)
Calculation • Multiply the mass of each isotope by its abundance as a decimal • Add each of these to get weighted average • Try one
Mass Spectrometer • Inject gaseous form of element • Strip electrons (positive charge) • Sort by size with a magnetic field • Computer counts the isotope and gives a readout
Animation • http://www.colby.edu/chemistry/OChem/DEMOS/MassSpec.html