Part A: Atomic Structure
Part A: Atomic Structure . +. Protons. Neutrons. -. Electrons. 5. What element is represented by the diagram? ____________ . Boron. Soaps. Glass Manufacturing. Flare Guns. Ceramics. Fiberglass. Part B: Atomic Calculations . 6. Label the information provided in the periodic table. .

Part A: Atomic Structure
E N D
Presentation Transcript
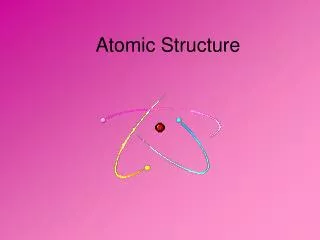
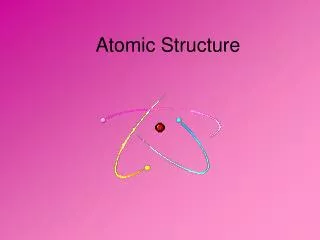
Part A: Atomic Structure + Protons Neutrons - Electrons
5. What element is represented by the diagram? ____________ Boron Soaps Glass Manufacturing Flare Guns Ceramics Fiberglass
Part B: Atomic Calculations 6. Label the information provided in the periodic table. Atomic Number 8 Symbol O Name Oxygen 15.999 Atomic Mass
7.What does the atomic number represent? Protons Electrons
What does the atomic mass represent? Protons + Neutrons Atomic Mass
9. How would you figure the number of protons or electrons in an atom? USE THE ATOMIC NUMBER Atomic Number 8
10. How would you figure the number of neutrons in an atom? SUBTRACT THE ATOMIC NUMBER FROM THE ATOMIC MASS Atomic Number 8 15.999 Atomic Mass
11. Use your knowledge of atomic calculations to complete the chart. 4 3 3 15 16 15 17 18 17 28 59 28 19 19 20 108 47 47 1 1 0 28 14 14 184 74 74 10 20 10
Part C: Electron Configuration 12. How many electrons can each level hold? 8 18 2 1st = 2nd = 3rd =
13. What term is used for the electrons in the outermost shell or energy level? VALENCE
Scientists use two types of diagrams to show the electron configuration for atoms. Bohr Diagram Lewis Structure
Bohr Diagram Shows all electrons Sulfur • Atomic # = 16 • Atomic Mass = 32 • Protons = • Neutrons = • Electron = 16 16 16
Lewis Structure Shows valence electrons Sulfur Atomic # = 16 Atomic Mass = 32 Protons = 16 Neutrons = 16 Electron = 16 S
Li • Atomic Number 3 • Mass Number 7 • Number of Protons • Number of Neutrons • Number of Electrons 3 4 3 Li
Ne • Atomic Number 10 • Mass Number 20 • Number of Protons • Number of Neutrons • Number of Electrons 10 10 10 Ne
Mg • Atomic Number 12 • Mass Number 24 • Number of Protons • Number of Neutrons • Number of Electrons 12 12 12 Mg
Cl • Atomic Number 17 • Mass Number 35 • Number of Protons • Number of Neutrons • Number of Electrons 17 18 17 Cl
He • Atomic Number 2 • Mass Number 4 • Number of Protons • Number of Neutrons • Number of Electrons 2 2 2 He
Si • Atomic Number 14 • Mass Number 28 • Number of Protons • Number of Neutrons • Number of Electrons 14 14 14 Si
(1) Which elements had a filled outermost shell? (2) Which element would be most likely to lose electrons in a chemical bond? (3) Which element would be most likely to gain electrons in a chemical bond? (4) Which elements are not likely to bond with other elements? Why? He & Ne Li (Only has 1 valence electron) Cl (Only needs 1 more electron to fill its outer shell) They have full outer shells. He & Ne