Fun with Phase Changes in Science Class
Explore chemical and physical changes through engaging activities and discussions on phase transitions like melting, freezing, vaporization, and more. Understand the science behind heat energy absorption and release.

Fun with Phase Changes in Science Class
E N D
Presentation Transcript
Sit quietly and begin your warm up.No writing on the lab tables!!!!!!!Chemical and Physical Changes is the agenda for today.Homework: Page 69 in the interactive noteboook.
Review: 3 Phases of Matter • Solid • Liquid • Gas
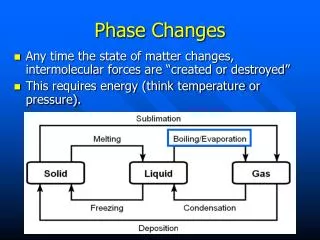
What is a Phase Change? • Is a change from one state of matter (solid, liquid, gas) to another. • Phase changes are physical changes because: - It only affects physical appearance, not chemical make-up. - Reversible
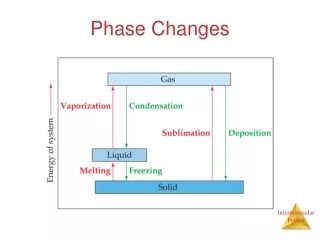
What happens during a phase change? • During a phase change, heat energy is either absorbed or released. • Heat energy is released as molecules slow down and move closer together. • Heat energy is absorbed as molecules speed up and expand.
Types of Phase Changes • Types of Phase Changes
Melting • Phase change from a solid to a liquid • Molecules speed up, move farther apart, and absorb heat energy
Freezing • Phase Change from a liquid to a solid • Molecule slow down, move closer together and release heat energy.
Vaporization (Boiling) • Phase change from a liquid to gas. It occurs at the boiling point of matter. • Molecules speed up, move farther apart, and absorb heat energy.
Evaporation • Phase change from a liquid to a gas on the surface of a liquid (occurs at all temperatures). • Molecules speed up, move farther apart, and absorb heat energy.
Condensation • Phase change from a gas to a liquid. • Molecule slow down, move closer together and release heat energy.
Sublimation • Phase change from a solid to a gas. • Molecules speed up, move farther apart, and absorb heat energy.
Deposition • Phase change from a gas to a solid. • Molecules slow down, move closer together and release heat energy.
Melting & Boiling Points • Melting Point: The temperature at which a solid changes into a liquid. • Boiling Point: The temperature at which a liquid changes into a gas. • What is a Freezing point? Compare the freezing and melting points of water.
Why is there no change in temperature during a phase change? Define melting and boiling points. What is the melting point and boiling point of water? At what temperature does water freeze and become a solid? Graphing a Phase Change