Periodic Table
Periodic Table. Larry Scheffler Lincoln High School. The Periodic Table-Key Questions. What is the periodic table ? What information is obtained from the table ? How can elemental properties be predicted based on the Periodic Table ?.

Periodic Table
E N D
Presentation Transcript
Periodic Table Larry Scheffler Lincoln High School
The Periodic Table-Key Questions • What is the periodic table ? • What information is obtained from the table ? • How can elemental properties be predicted based on the Periodic Table?
The development of the periodic table brought a system of order to what was otherwise an collection of thousands of pieces of information The periodic table is a milestone in the development of modern chemistry. It not only brought order to the elements but it also enabled scientists to predict the existence of yet undiscovered elements. Periodic Table
Early Attempts to Classify Elements • Dobreiner’s Triads (1827) • Classified elements in sets of three having similar properties. • Found that the properties of the middle element were approximately an average of the other two elements in the triad.
Dobreiner’s Triads Note: In each case, the numerical values for the atomic mass and density of the middle element are close to the averages of the other two elements
Newland’s Octaves -1863 • John Newland attempted to classify the then 62 known elements of his day. • He observed that when classified according to atomic mass, similar properties appeared to repeat for about every eighth element • His Attempt to correlate the properties of elements with musical scales subjected him to ridicule. • In the end his work was acknowledged and he was vindicated with the award of the Davy Medal in 1887 for his work.
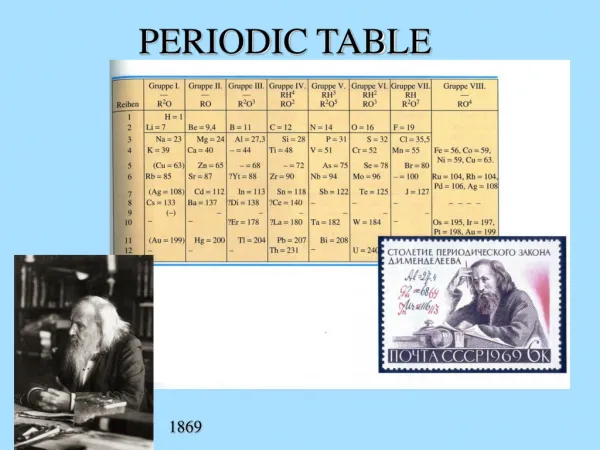
Dmitri Mendeleev Dmitri Mendeleev is credited with creating the modern periodic table of the elements. He gets the credit because he not only arranged the atoms, but he made predictions based on his arrangement which were shown to be quite accurate.
Mendeleev’s Periodic Table • Mendeleev organized all of the elements into one comprehensive table. • Elements were arranged in order of increasing mass. • Elements with similar properties were placed in the same row.
Mendeleev’s Periodic Table Mendeleev left some blank spaces in his periodic table. At the time the elements gallium and germanium were not known. He predicted their discovery and estimated their properties
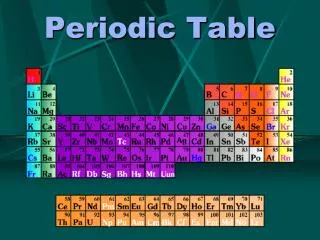
Periodic Table The Periodic Table has undergone several modifications before it evolved in its present form. The current form is usually attributed to Glenn Seaborg in 1945
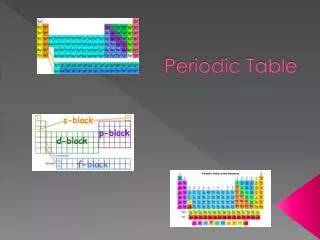
Periodic Table Expanded View • The Periodic Table can be arranged by energy sub levels The s-block is Group IA and & IIA, the p-block is Group IIIA - VIIIA. The d-block is the transition metals, and the f-block are the Lanthanides and Actinide metals • The way the periodic table usually shown is a compressed view. The Lanthanides and actinides (F block)are cut out and placed at the bottom of the table.
PeriodicTable: Metallic Arrangement • Layout of the Periodic Table: Metals vs. nonmetals Nonmetals Metals
The Three Broad Classes Are Main, Transition, Rare Earth • Main (Representative), Transition metals, • lanthanides and actinides (rare earth)
Reading the Periodic Table: Classification • Nonmetals, Metals, Metalloids, Noble gases
Periodic Table: The electron configurations are inherent in the periodic table • B • 2p1 H 1s1 He 1s2 F 2p5 Be 2s2 B 2p1 C 2p2 N 2p3 Ne 2p6 O 2p4 Li 2s1 Na 3s1 Mg 3s2 Cl 3p5 Si 3p2 S 3p4 Ar 3p6 Al 3p1 P 3p3 K 4s1 Ca 4s2 Zn 3d10 As 4p3 Be 4p5 V 3d3 Mn 3d5 Fe 3d6 Co 3d7 Sc 3d1 Ti 3d2 Ga 4p1 Ge 4p2 Se 4p4 Cr 4s13d5 Kr 4p6 Ni 3d8 Cu 4s13d10 Sr 5s2 Rb 5s1 Nb 4d3 Ru 4d6 Rh 4d7 Mo 5s14d5 Cd 4d10 Sn 5p2 I 5p5 Xe 5p6 Zr 4d2 Tc 4d5 Y 4d1 In 5p1 Sb 5p3 Te 5p4 Ni 4d8 Ag 5s14d10 Hf 5d2 Cs 6s1 Ta 5d3 Re 5d5 Os 5d6 Ir 5d7 W 6s15d5 La 5d1 Rn 6p6 At 6p5 Ni 5d8 Ba 6s2 Hg 5d10 Tl 6p1 Pb 6p2 Bi 6p3 Po 6p4 Au 6s15d10 Mt 6d7 Bh 6d5 Hs 6d6 Fr 7s1 Rf 6d2 Ra 7s2 Db 6d3 Sg 7s16d5 Ac 6d1
Periodic Table Organization------ Groups or Families Vertical columns in the periodic table are known as groups or families The elements in a group have similar electron configurations
Periodic Table Organization ---- Periods Horizontal Rows in the periodic table are known as Periods The Elements in a period undergo a gradual change in properties as one proceeds from left to right
Periodic Properties • Elements show gradual changes in certain physical properties as one moves across a period or down a group in the periodic table. These properties repeat after certain intervals. In other words they are PERIODIC Periodic properties include: -- Ionization Energy --Electronegativity -- Electron Affinity -- Atomic Radius -- Ionic Radius
Trends in Ionization Energy Ionization energy is the energy required to Remove an electron from an atom • Ionization energy increases across a period because the positive charge increases. • Metals lose electrons more easily than nonmetals. • Nonmetals lose electrons with difficulty (they like to GAIN electrons).
Trends in Ionization Energy • The ionization energy increases UP a group • Atoms near the top of a group are effected less by the Shielding Effect.
Electronegativity Electronegativity is a measure of the ability of an atom in a molecule to attract electrons to itself. This concept was first proposed by Linus Pauling (1901-1994). He later won the Nobel Prize for his efforts
Periodic Trends: Electronegativity • In a group: Atoms with fewer energy levels can attract electrons better (less shielding). So, electronegativity increases UP a group of elements. • In a period: More protons, while the energy levels are the same, means atoms can better attract electrons. So, electronegativity increases RIGHT in a period of elements.
The Electron Shielding Effect • Electrons between the nucleusand the valence electrons repel each other making the atom larger.
Atomic Radius • The radius increases down a group. • Because electrons are added further from the nucleus, there is less attraction. This is due to additional energy levels and the shielding effect. Each additional energy level “shields” the electrons from being pulled in toward the nucleus. • The radius decreases on going across a period.
Atomic Radius The radius decreases across a period owing to increase in the positive charge from the protons. Each added electron feels a greater and greater + charge because the protons are pulling in the same direction, where the electrons are scattered. Large Small
Core Charge • The concept of core charge can be used to better understand trends in atomic radii. • Core charge = nuclear charge (ie: number of protons) – number of inner-shell electrons. • Eg: Na (s) 11p – 10e = +1
Core Charge • The greater the core charge, the greater the attraction of protons to electrons. • greater core charge means SMALLER atomic radii. • Core charge increases across period 3 so atomic radii decreases.
Trends in Ion Sizes Radius in pm
Cations Cations (positive ions) are smaller than their corresponding atoms
Ion Sizes Does the size go up or down when gaining an electron to form an anion?
+ + Li , 78 pm 2e and 3 p Ionic Radius Forming a cation. • CATIONS are SMALLER than the atoms from which they come. • The electron/proton attraction has gone UP (increased core charge) and so the radius DECREASES. Li,152 pm 3e and 3p
Ionic Radius for Cations Positve ions or cations are smaller than the corresponding atoms. Cations like atoms increase as one moves from top to bottom in a group.
Anions Anions (negative ions) are larger than their corresponding atoms
- - F 64 pm F , 133 pm 9e- and 9p+ 10 e- and 9 p+ Ionic Radius-Anions • ANIONS are LARGER than the atoms from which they come. • The electron/proton attraction has gone DOWN (core charge decreases) and so size INCREASES. • Trends in ion sizes are the same as atom sizes. Forming an anion.
Ionic Radii for Anions Negative ions or anions are larger than the corresponding atoms. Anions like atoms increase as one moves from top to bottom in a group.
Ionic Radius for an Isoelectronic Group Isoelectronic ions have the same number of electrons. The more negative an ion is the larger it is and vice versa.
The Periodic Table--Summary The periodic table is a classification system. Although we are most familiar with the periodic table that Seaborg proposed more than 60 years ago, several alternate designs have been proposed.