The Iron
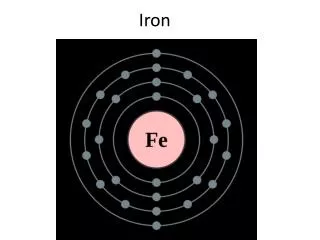
The Iron. Generalities. S y mbol : Fe. 26 p + , 26 e - , the 26th element in Periodic System. A= Z + n o. n o = 56 – 26 n o = 30. Nuclear task is + 26. A= 56 => 1 mole Fe = 56 g. => 1 kmole Fe = 56 kg.

The Iron
E N D
Presentation Transcript
Generalities Symbol : Fe 26 p+, 26 e-, the 26th element in PeriodicSystem A= Z + no no= 56 – 26 no= 30 Nuclear task is + 26. A= 56 => 1 mole Fe = 56 g. => 1 kmole Fe = 56 kg.
- The ironis the 10th most common element in Universeand the 4th on Earth (2nd metal after aluminium).
Irons positive ions gives away + 2 Fe Fe -2 e- divalent positive ion gives away +3 Fe Fe -3 e- trivalent positive ion
Natural state It’s natural state is only in meteorites ºIn nature he’s combined with other substances forming oxides, sulphates… ºIron is 4th most common element in Earths shell.
Obtaining methods By aluminothermia: t˚ 2 Al + Fe2O3 Al2O3 + 2Fe + +
Physical properties • solid; • -grey; • doesn’t dissolve in water; • thermical and electrical conductor; • special magnetic properties; • pliable;
Chemical properties of Fe Non-metals ( O2, S) Reacts with: Metals (Al )
Fe reaction with non-metals: t˚ (yellow sparks). FeO2 Fe + O2 t˚ +
t˚ 2FeCl3 2Fe +3Cl2 t˚ +
Reaction with Sulfur Fe + S FeS
Reaction of Fe with H2O In pure water, Fe doesn’t rust. In drinking water Fe rusts. When we heat up iron: t˚ 3Fe + 4H2O(v) Fe3O4 + 4 H2
The reaction of iron with acids Fe + HCl 2 FeCl2 + H2
Fe + H2SO4 FeSO4 +H2 Sulfuric acid
The reaction of iron with salts Fe + CuSO4 FeSO4 + Cu ↓
Uses of Iron • Electromagnets • Wires • Corrugated iron • HNO3 Tanks • Pipes and vessels • Radiators and stoves
The End This movie was made by: Sitaru Andreea Leanca Daniela Cojocaru Oana-Elena Teacher : Amazilitei Adriana Other information sources: Chemistry Book (VIIIth grade)