Effective Atomic Bonding: Cornell Notes and Beyond
Learn Cornell Note-taking method for atomic bonding, types of bonds (Covalent, Ionic), and periodic table insights with effective study tips. Improve synthesis and knowledge application.

Effective Atomic Bonding: Cornell Notes and Beyond
E N D
Presentation Transcript
Quick Tip • Any slides that appear in this pattern are the slides you need to copy in your notes (Cornell note style). • Any text that is italicized is important and should be written down in your notes. • All other slides and text, do not need to be written down. • Any questions?
Cornell Notes? • The paper gets divided into 1/3 and 2/3 sections. Leaving space at the top and bottom.
Cornell Notes • Name, Class/Period, Date go in the upper right corner. • Topic/Objective/vocabulary(written after the notes are taken) in the upper left. • Essential question goes here.
Cornell Notes • Questions and key points go on the left. • Class notes go on the right. • A review summary is located at the bottom.
The Cornell method • systematic format for condensing and organizing notes • main ideas of the text or lecture • long ideas are paraphrased • long sentences are avoided • symbols or abbreviations are used instead • relevant questions or key words are written in the key word column • Within 24 hours of taking the notes, the student must revise and write questions and then write a brief summary in the bottom five to seven lines of the page This helps to increase understanding of the topic • Helps when studying for either a test or quiz • When reviewing the material, the student can cover the note-taking (right) column while attempting to answer the questions/keywords in the key word or cue (left) column.
Effectiveness • A study published in 2008 by Wichita State University compared two note taking methods: • Cornell and Guided. • They found that Cornell Note taking is more effective for students improvement of synthesis and apply learned knowledge • While the guided notes method appeared to be better for basic recall. • Basically • Cornell notes make you a thinker • Guided make you a repeater.
Let’s Review What We Know • What are the three parts to the atom? • What are the charges on these particles? • Where in the atom is each particle located?
Cornell Notes • Objectives • Define Covalent and Ionic bonds. • Interpret the periodic table and predict the type of ion that will form.. • Draw a model of an ionic bond.
Cornell Notes • Essential Question • What are the types of scientific bonds and how can I use a model to represent each?
The Periodic Table • Are there any similarities or patterns? H He Li Be B C N O F Ne Na Mg Al Si P Cl S Ar
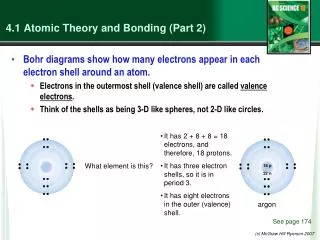
Periodic Table • What is a valence electron? • A valence electron is any electron or electrons on the outermost energy level. • Each atom has between 1 and 8 valence electrons.
Periodic Table • The periodic table can quickly tell you how many valence electrons an element has according to where it is located. • Location can also tell you how many energy levels there are. • How?
Periods and Groups • Periods are the row that each element is located in and tells you how many energy levels there are. • Groups are the column that each element is in and tells you the number of valence electrons. • IMPORTANT: the group rule does not apply to the transition metals located in the center of the periodic table. Which is not represented on the table you created.
Periods and Groups Example 1 2 3 4 5 6 7 8 1 2 3 H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar
Quick Activity • Take five minutes to complete this activity • Predict the energy levels for: • Potassium, Bromine, and Radon • Predict the number of valence electrons for: • Radium, Arsenic, and Iodine • Periodic Table
Activity Answers • Energy levels: • Potassium (K)– 4 • Bromine (Br) – 4 • Radon (Rn)- 6 • Valence Electrons: • Radium (Ra) – 2 • Arsenic (As) – 5 • Iodine (I) - 7
Moving on to Bonding • Let’s look at our old familiar friend: • The water molecule. • What is the water molecule made of, what elements? • 2 Hydrogen and 1 oxygen. • Or let’s take a look at our everyday edible companion: • Table salt NaCl.
Bonding • H2O • NaCl O H H Na+ Cl-
Bonding • What makes them stick together? • What makes them Bond? • What does it mean to bond? • When a marriage occurs, it is said that there is a bond made between the bride and groom. • Same thing with elements when they become close or make a connection then a bond occurs.
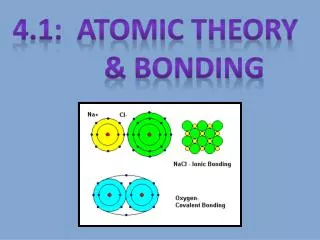
Bonding • When two or more atoms join together to make a molecule. • There are three types of bonds (we will only discuss the first two): • Ionic- Bond between oppositely charged ions. • Covalent- Bond when atoms share one or more pairs of electrons. • Metallic- bond formed between positively charged metal ions and the electrons around them.
Ionic Bonding • What can you tell me about the protons and electrons in an atom? • In a neutral atom the number of electrons matches the number of protons. • What are their charges? • Electrons are negative and protons are positive.
Losing Particles • An atom can never, never, lose or gain a proton(s) on its own. • If it did it would: • Change the element it is • Release so much energy that it would result in a nuclear explosion • An atom can lose or gain electron(s) on its own.
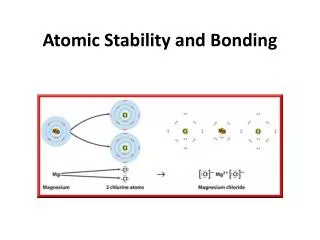
For Example • Let’s use magnesium as an example: • How many protons does a neutral atom of Magnesium have? • 12 • How many electrons? • 12 • The positives balance the negatives. Each negative has a positive to match it. + + + + + + + + + + + + - - - - - - - - - - - -
Neutral Atom • When the positive charge is balanced by the negative charge in an atom, the atom is neutral. There is no over all net charge. + + + + + + + + + + + + • - - - - - - - - - - - • Each proton in magnesium has a matching electron, so: -12 + (+12) = 0 net charge • This is true for all neutral atoms. The positives cancel out the negatives.
Ionic Bonding - Losing Electron Example If our Magnesium atom loses an electron, what happens to its charge? + + + + + + + + + + + + - - - - - - - - - - - ? • The positive charges and negative charges no longer balance out. There will be more positive charges than negative charges resulting in a positive net charge. -11 (lost an electron) + (+12) = +1 net charge
Ionic Bonding - Losing Electron Example, too. • What if our Magnesium atom lost two electrons? + + + + + + + + + + + + - - - - - - - - - - ? ? -10 (lost two electrons) + (+12) = +2 net charge
Ionic Bonding - Gaining Electron Example What happens to the charge of Oxygen if it gains an electron? + + + + + + + + ? - - - - - - - - - • The positive charges and negative charges no longer balance out. There will be more negative charges than positive charges resulting in a negative net charge. -9 (gain an electron) + (+8) = -1 net charge
Ionic Bonding - Gaining Electron Example, too. What happens to the charge of Oxygen if it gains two electron? + + + + + + + + ? ? • - - - - - - - - - -10 (Gained two electrons) + (+8) = -2 net charge
Ionic Bonding - Losing or Gaining Electrons • If an atom is losing electrons: • It will always be positive. • It will never lose more electrons than it has on its valence shell. • If an atom is gaining electrons • It will always be negative. • It will never gain more electrons that it takes to fill the valence shell.
Ionic Bonding So why did we take all that time to go over gaining and losing? Because…. • This will explain what an ion is. • Ion- an atom or group of atoms that has lost or gained one or more electrons and has a negative or positive charge. It is no longer a neutral atom. • removing or adding an electron from an atom an ion is created. • Atoms like to have complete outer shells, eight electrons on their outer shell (octet rule). (or two if there is only one shell) • Only a few atoms (elements) have a complete outer shell. So all the others have to gain or lose electrons. • Knowing this, would it be easier to gain or lose electrons, and why?
Ionic Bonding • Atoms in group 1 are very reactive and as such are very willing to give up their electron in order to have a complete shell. • What about the atoms in group 7? Would they gain or give up an electron? • They are more likely to gain an electron, why? • It’s easier to gain one electron than give up seven.
Positive and Negative Ions • Atoms of the metal elements (Boron included), such as sodium and calcium, form the positive ions.(columns 1, 2, 3) • Atoms of the non-metals, such as chlorine and oxygen, form the negative charged particles. (columns 5, 6 and 7) • But what about column 4? What ions do they form? • They can form either positive (lose electrons) or negative (gain electrons) ions.
Ionic Bonding • Sodium Chloride is made from sodium and chloride ions. • What do you think the charge is on the sodium ion? What made you come to your decision? • How about the chloride ion?
Ionic Bonding • So we have Na+ and Cl- ions. • What does this all have to do with bonding? • Ionic bonds occur between oppositely charged ions.
Ionic Bonding • Ionic Bonds are formed by the transfer of electrons between a metal and a non-metal atom. • One atom gains the electron(s) that the other atom loses. • This results in a positive ion (cation) and a negative ion (anion). • Because one atom is positive and the other is negative the positive ion attracts the negative ion to form a bond.
Ionic Bonds • What is electron affinity? • the power that the positive nucleus of an atom has to attract the electrons of another atom. • How does that relate to ionic bonds? • If the affinity is much greater in one atom than the other then the atom with the greater affinity has the ability to steal the electron creating an ionic bond. • Ionic bonds are stronger than covalent due to their higher electron affinity.
Ionic Bonding • These attractions eventually form a network of bonded ions. • This network then forms crystals, in our NaCl case, crystals of table salt. • This compound NaCl is made from a positive and negative ion, resulting in a neutral compound of NaCl. • One positive Na ion balances one negative Cl ion. • So there is a 1:1 ratio, 1 Na to 1 Cl.
What is Ionic charge? • Another name for net charge • The charge of an atom when you add the number of protons to the number of electrons. • Ionic charge represents the number of electrons an element will gain or lose.
Ionic/Net Charge • Neutral atoms have no net charge • Positive ions have more protons than electrons and a positive net charge • Cation – positively charged ion • Negative ions have more electrons than protons and a negative net charge. • Anion- negatively charged ion
Examples • Example: • Lithium • Since lithium is in column 1 it needs to lose 1 electron to have a complete shell so it will have an ionic charge of +1 • Magnesium • Since Magnesium is in column 2 it needs to lose 2 electrons to have a complete shell so it will have an ionic charge of +2
Examples Continued. • Boron • Since boron is in column 3 it needs to lose three electrons to have a complete outer shell so it will have an ionic charge of +3 • Fluorine • Since fluorine is in column 7 it needs to gain an electron to have a complete shell so its ionic charge would be -1
Examples Continued • Oxygen • Since oxygen is in column six it needs to gain two electrons to have a complete shell so its ionic charge would be -2 • Nitrogen • Since Nitrogen is in column 5 it needs to gain 3 electrons to have a full shell so its ionic charge would be -3
A Special Case Hydrogen • Hydrogen can gain or lose 1 electron so it can have an ionic charge of -1 if it gains and +1 if it loses.
How do I symbolize an Ion? • When writing the shorthand for an Ion • You need to include the symbol and charge. • the symbol is written normal with the charge written as superscript in the upper right “corner” of the atomic symbol. • Oxygen’s ion is written O2+ • Hydrogen’s ion is written H1+or H1-
Ionic Bonding Activity • Figure out the ions for Aluminum and Phosphorous.
Ionic Bonding Activity Solution • First you have to decide what ions form. • For Aluminum: • Will it be positive or negative? • What is the value of charge? • For Phosphorous • Will it be positive or negative? • What is the value of the charge?
Ionic Bonding Activity Solution • Aluminum is in group 3. • How many valence electrons does it have? • So is it easier to give up or gain ? • So what would Aluminum’s ionic charge be, positive or negative? • If it gives up electrons it becomes positive. • So the ion is: Al3+. • Aluminum gives up 3 electrons allowing the protons to outnumber the remaining electrons by three.