Atomic Bonding
Explore atomic bonding, matter composition with elements, mole calculations, and the essential role of quantum mechanics in the Bohr model. Learn about gold nanoparticles, stained glass, and quantum properties in materials science.

Atomic Bonding
E N D
Presentation Transcript
Atomic Bonding Chapter 2
Matter is composed of Elements • 92 naturally occurring • Atomic Number • # of protons • Mass Number • # of protons + # of neutrons • Atomic Weight • Average • Grams/mole
Mole • # of atoms in 12 grams of Carbon-12 • Avogadro’s Number • 6.022*10^23 • lb mole used in English calculations • # of atoms in 12 lbm of Carbon-12 • What kind of calculations can you do using the definition of a mole?
Bohr Model of the Atom • Both Bohr and Schrödinger knew that matter can be thought of as either a particle or a wave • Bohr chose to think of electrons as particles with wave characteristics • This approach is usually more intuitively easy to understand
Practically • Most things we do in Materials Science can be handled with the Bohr model • However… • Understanding the behavior of semiconductors requires some elementary understanding of quantum mechanics • Exciting advances are being made using Nanomaterials for which the Bohr model is inadequate – we need quantum mechanics
For Example… • Gold has interesting properties when the particles are very small – on the nanoscale • In order to understand this behavior, you need quantum mechanics
Gold has been used since medieval times to create red glass Stained glass in Sulkowski castle in Bielsko-Biała , Poland European panel, 1564, with typical painted details, extensive silver stain, Cousin's rose on the face and flashed ruby glass with abraded white motif http://en.wikipedia.org/wiki/Stained_glass
Other Colors • Copper oxides were added to produce green, • Cobalt for blue, and • Gold was added to produce red glass.
Nasir ol Molk mosque, Qajar era, Shiraz. Iran http://en.wikipedia.org/wiki/Image:Nasirolmolk.jpg
Sainte-Chapelle in Paris: Rose window. http://en.wikipedia.org/wiki/Image:Sainte-Chapelle-Rose-window.jpg
Why? • Gold on the bulk scale is yellow Placer Gold - Flattened nuggets of native gold found in Lynx Creek, Arizona. Gold in quartz http://www.gc.maricopa.edu/earthsci/imagearchive/gold.htm
Gold Nanoparticles are red • The wavelengths of light that are absorbed change for nanoparticles • In very small particles there are so few atoms that the interactions are affected by quantum effects
Gold Nanoshells • When you coat a glass (SiO2) particle with gold it is called a nanoshell Computer simulation of the growth of gold nanoshell over a silica core http://www.genomenewsnetwork.org/articles/07_03/nanoshells.shtml
Colloidal gold on the left – Gold nanoshells tuned to absorb different wavelengths of light (6 vials on the right) http://www.nanospectra.com/physics/physics.asp
Naomi Halas http://www.ece.rice.edu/~halas/halas.html
Nova Now http://www.pbs.org/wgbh/nova/sciencenow/3209/03.html
Clinical Trials • Currently underway on “refractory” head and neck tumors • http://www.nanospectra.com/index.html
Quantum Mechanics • What the Bohr model can’t explain is that only certain energy states are possible for an electron – the very property that makes gold nanospheres so interesting • Schrödinger thought of the electron as a wave - that sometimes behaves like a particle
Other wavelengths are impossible if each end is held in place
Electrons can be thought of as standing waves • Only certain energy levels are possible • Wave particle duality Violin strings vibrate in a standing wave
We model many things using mathematics • For example, the equation of a line is • y=a*x + b • a stands for the slope • b stands for the intercept
We model an electron as a wave, described with an equation with four parameters, the quantum numbers • n • l • ml • ms
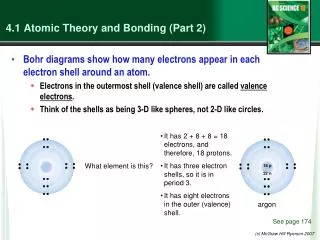
Primary Quantum Number • n • Can have values from 1 to infinity, but they can only be integers • K, L, M, N • Represents the energy of the orbital, which is also related to the size of the orbital • An orbital is the region of space where you are likely to find the electron
Angular Momentum Quantum Number • l • Shape of the orbital • Can have values from 0 to n-1 • s, p, d, f, g, h….. • If there is more than one electron present, the angular momentum quantum number also affects the orbital energy (also called the azimuthal quantum number)
Magnetic Quantum Number • ml • Can have integer values from –l to +l • Thus, if n=1, l =0, and mlmust equal 0 • In other words, it can only have one value • If n=2, then l can equal either 0 or 1 • If it equals 1, then ml can equal –1, 0 or +1 • It can have three values
Remember • S orbitals correspond to l = 0 • P orbitals correspond to l = 1 • D orbitals correspond to l = 2 • F orbitals correspond to l = 3 • How many orbitals are possible for each of these types? 1 3 5 7 Corresponds roughly to the orientation of the orbital in space
http://www.alevelchemistry.co.uk/Module_1/HTML_Pages/Atomic_Structure/1.1_Atomic_Structure_Notes.htmhttp://www.alevelchemistry.co.uk/Module_1/HTML_Pages/Atomic_Structure/1.1_Atomic_Structure_Notes.htm
Spin Quantum Number • ms • +1/2 • -1/2 • Two electrons of opposite spin fill each orbital
The first three quantum numbers define an orbital • You need all four to define an electron
Shorthand Notation • Germanium has 32 protons and 32 electrons • 1s22s22p63s23p63d104s24p2
Electron Shells • Bonding occurs only with the electrons in the outer most shells – called the valence electrons
Inner electrons are called the core electrons • The valence electrons are those in the outer s and p orbitals, and any unfilled d and p orbitals.
1s22s22p63s23p63d104s24p2 Germanium Valence electrons Core electrons 1s22s22p63s23p63d24s2 Titanium Chemistry happens in the valence shell
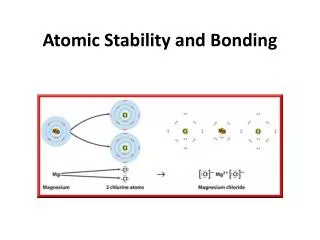
Electronegativity • The tendency of an atom to gain an electron • The most stable configuration of electrons is completely filled valence shells
- Cl Non-Metals • Elements to the right side of the periodic table • The easiest way to achieve a full valence shell is to add electrons • They have a high electronegativity Readily form anions
Na Metals • Elements to the left of the periodic table • The easiest way to achieve a full valence shell is to give up electrons • They have a low electronegativity Na+ Readily form cations
Elements with intemediate properties – usually considered poor metals
Electronegativity controls how elements combine (bond) with each other.
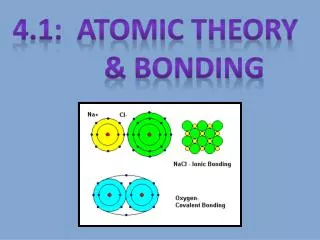
Atomic Bonding • Metallic • Ionic • Covalent
Metallic Bonds • Metal-Metal • Non-directional • Electrons are free to move around • Good electrical conductivity • Good thermal conductivity • What is a molecule? Metals are formed from metallic bonds
Metallic Bonding http://www.chm.bris.ac.uk/pt/harvey/gcse/other.html
Ionic Bonds • Metal-Nonmetal • Cation-anion • Non-directional • Poor electrical conductivity • Poor thermal conductivity • Ceramics are formed from ionic bonds • What is a molecule?
Ionic Bonding http://ibchem.com/IB/ibfiles/bonding/bon_img/NaCl4.jpg