Atomic Structure and Bonding
Atomic Structure and Bonding. The Structure of Atoms. Specific Learning Outcomes. Describe or label a diagram of a model of an atom using the terms protons, neutrons, electrons and nucleus. Define atomic number (Z) Define mass number (A) Define and give examples of isotopes.

Atomic Structure and Bonding
E N D
Presentation Transcript
Atomic Structure and Bonding The Structure of Atoms
Specific Learning Outcomes • Describe or label a diagram of a model of an atom using the terms protons, neutrons, electrons and nucleus. • Define atomic number (Z) • Define mass number (A) • Define and give examples of isotopes. • Calculate the number of electrons, protons and neutrons in any named atom. • Write electron configurations of the first 20 elements using 2,8,8 notation.
A little Atomic History In 1806 John Dalton reintroduced the idea of an atom. His theory suggested that all elements were made up of atoms. These atoms were unique to the element and could not be created or destroyed. When atoms from different elements joint together they would form compounds. Jons Berzelius 1813 Devised the “Chemical Symbol” First letter uppercase, second letter lower case. J J Thomson 1897 Discovered electrons. Ernest Rutherford 1911 Discovered a lot more about atomic structure – like polarity and density. AmedeoAvogrado 1811 Discovered that some elements existed in combinations atoms like Cl2 or O2 Henry Moseley 1914 Learnt how to count the positive charges in an atom and how electrons were arranged.
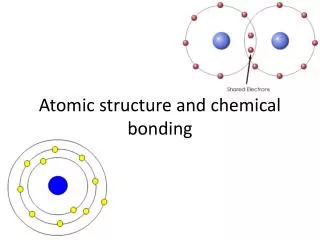
The Structure of Atoms Electron (Negative Charge) Neutron (No Charge) Proton (Positive Charge) Nucleus Energy Level • There are three pieces to an atom. There are electrons, protons, and neutrons. • As you know, there are over 100 elements in the periodic table. The thing that makes each of those elements different is the number of electrons, protons, and neutrons. • The protons and neutrons are always in the centre of the atom. Scientists call the centre of the atom the nucleus. • The electrons are always found whizzing around the centre in areas called orbitals.
Atomic Number and Mass Number Chemists have a symbolic way of representing atoms which enables them to quickly see how many protons, neutrons and electrons an atom has… Mass Number Element Symbol Atomic Number Mass Number = Number of protons plus neutrons in the nucleus Atomic Number = Number of protons in the nucleus or Number of electrons outside of the nucleus
Isotopes Atoms of the same element can have different numbers of neutrons; the different possible versions of each element are called isotopes. Hydrogen Deuterium Tritium For example, the most common isotope of hydrogen has no neutrons at all; there's also a hydrogen isotope called deuterium, with one neutron, and another, tritium, with two neutrons.
Atomic Number and Mass NumberActivities What's the most important thing to learn in chemistry?Never lick the spoon.
Electron Configuration An electron configuration of an atom is a description of the number of electrons in each energy level. Whatever is left over 2 8 8 ? 4th Level Nucleus 1st Level 2nd Level 3rd Level To complete an electron configuration take the atomic number (Z) and spread it out through the levels – each level has a maximum number of electrons that it will hold as demonstrated above. For example for the element calcium where Z = 20, the electron configuration would be as 2,8,8,2.