The Evolution of Atomic Theory and the Development of the Periodic Table
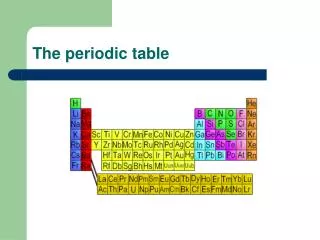
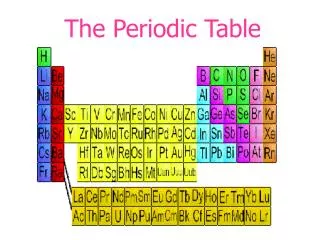
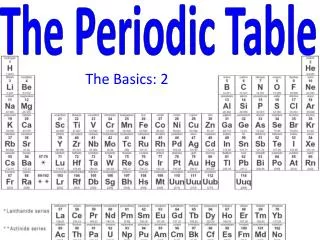
The Periodic Table, established in 1868 by Dmitri Mendeleev, was organized based on the properties of known elements, revealing a periodicity in their characteristics such as melting point and density. Mendeleev, unaware of protons and electrons, collected elemental data and identified gaps in the table, accurately predicting properties of undiscovered elements like Gallium and Germanium. This document also outlines the historical milestones in atomic theory, from Democritus’s early concepts to the modern quantum theory, highlighting pivotal contributions from Dalton, Thomson, Rutherford, and Bohr.

The Evolution of Atomic Theory and the Development of the Periodic Table
E N D
Presentation Transcript
The Periodic Table • 1868 • Was organized according to the properties of the known elements • Dmitri Mendeleev noticed that when the elements were put into order by their atomic mass, some of their properties repeated periodically
Dmitri Mendeleev • Noticed that properties repeat, e.g. melting point, react with oxygen, density, mass…
Periodic Table • So he put them in columns with others that are similar. • Didn’t know anything about protons, electrons, etc. Just made observations! • Even made some guesses about ones that were missing and their properties. He was correct!
Guesses • Eka-Aluminum: • Atomic Mass 68, density 5.9 • Gallium (Ga): • Atomic mass 69.7, density 5.94 • Eka-Silicon: • Atomic Mass 72, density 5.5 • Germanium (Ge): • Atomic Mass 72.6, density 5.7
Atomic Theory • Democritus …5th century BCE • Matter is made up of atoms • Atoms are solid • No internal structure • Different sizes • Space in between them (void)
Atomic Theory • John Dalton … 1800’s • Father of modern atomic theory • Discovered atomic weights and symbols for elements • elements made of atoms • atoms of an element same mass • different elements have different masses • Atoms combine in small, ratios such as 1:1, 1:2, 2:3 • atoms can’t be created or destroyed
Atomic Theory • JJ Thompson … 1897 • Discovered electron (first subatomic particle) • Believed atoms had negative charges floating in a sea of positive ones, like chocolate chips in a cookie • Called plum pudding model
Atomic Theory • Earnest Rutherford … 1911 • Discovered nucleus of atom and said it was positive • Electrons orbit through empty space around positive nucleus
Atomic Theory • Niels Bohr … 1914 • Discovered energy levels for electrons • Farther away from nucleus, higher energy level • Planetary model… like planets orbiting sun
Atomic Theory • Quantum theory • Current theory • Electrons don’t have orbits like planets • They move to fast to know exactly where they are, but we can tell where they probably are • Electron cloud is place where they probably are
Electron cloud • Broken down into energy levels • Energy levels have sublevels called orbitals • Energy levels and orbitals only hold a certain number of electrons maximum • Called s p d f