Plasmid maintenance
Plasmid maintenance. Maintaining plasmids and expressing those genes is energetically expensive to a bacterial cell. It is beneficial only if the bacteria obtains something useful from doing so.

Plasmid maintenance
E N D
Presentation Transcript
Plasmid maintenance Maintaining plasmids and expressing those genes is energetically expensive to a bacterial cell. It is beneficial only if the bacteria obtains something useful from doing so. As a result, bacteria will tend to lose plasmids when they are not beneficial. Bacteria that lose useless plasmids will have more energy to replicate faster than those that retain and express genes from unneeded plasmids and will out perform them over time. The process by which plasmids are lost is called curing. The rate at which plasmid curing occurs is determined by the conditions in which the bacteria resides. If conditions favor retention of the plasmid then there is selection for cells with plasmids, if there is no benefit for the plasmid, then there is selection for curing. In intermediate conditions cells may retain the plasmid at lower numbers or expression levels.
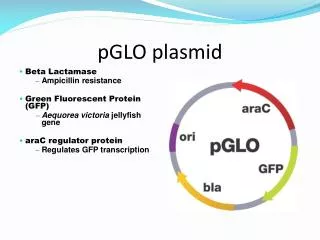
This week’s lab You will work as a group to perform a 5-day experiment to determine the rate of plasmid loss under permissive, selective, and intermediate conditions. You will work with a strain of E. coli bearing a high-copy plasmid expressing the Green Fluorescence Protein and resistance to ampicillin. Each lab will use bacterial cultures set up by a previous lab and will inoculate new cultures for later labs to continue the experiment. You will look at several things: CFU/ml of total bacteria and CFU/ml of bacteria bearing plasmid this will be done by plate counts on LB agar and LB agar plus ampicillin Fluorescence of bacterial cultures to determine rates of expression of GFP Plasmid DNA in bacterial cultures by isolating plasmid DNA from cultures and performing gel electrophoresis
This week’s lab 2 The results of each lab section will be collected and shared so you will see the results from one lab group for each lab section to compare to your own. You will analyze the data with respect to plasmid loss and reduced expression as a factor of time and selective pressure
Serial dilution and plate counts CFU=colony forming units. Number of cells capable of growth on plate Bacterial density is usually expressed as cfu/ml of suspension A dense bacterial suspension will have too many cells in each ml of fluid to count if plated, therefore, you make serial dilutions to obtain reasonable cell densities to count. Serial dilutions of 10-fold or 100-fold factor Plate a portion of the dilutions (usually 100-200µl) on a plate Count colonies on plates (looking for informative range of 20-250 colonies on one plate) Calculate cfu/ml of original suspension from this information
Calculation To calculate the bacterial density in the original suspension follow this calculation: In this example let’s assume that you had 32 colonies on a plate obtained by plating 100µl of a 1x10-6 dilution of the original suspension. First, determine the correction factor to adjust the volume plated on each plate to 1.0 ml. In our case since you plated 100µl you would need to multiply the number of colonies by 10 to determine how many colonies you would have gotten in 1.0 ml of the dilute suspension. In this cae 32 x 10=320. Next, you multiply by the inverse of the dilution factor. In our example you got an informative number of colonies from the 10-6 dilution, so we would multiply our adjusted number of colonies by 1x106 to correct for the dilution. 320 x 1x106 =320 x106 or 3.20x 108 cfu/ml in the original suspension.