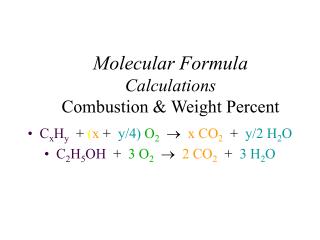Formula Calculations
Ch. 7 – The Mole. Formula Calculations. A. Percentage Composition. EMPIRICAL and MOLECULAR FORMULAS. the percentage by mass of each element in a compound. 127.10 g Cu 159.17 g Cu 2 S. 32.07 g S 159.17 g Cu 2 S. A. Percentage Composition. Find the % composition of Cu 2 S. 100 =.

Formula Calculations
E N D
Presentation Transcript
Ch. 7 – The Mole Formula Calculations
A. Percentage Composition EMPIRICAL and MOLECULAR FORMULAS • the percentage by mass of each element in a compound
127.10 g Cu 159.17 g Cu2S 32.07 g S 159.17 g Cu2S A. Percentage Composition • Find the % composition of Cu2S. 100 = %Cu = 79.852% Cu %S = 100 = 20.15% S
28 g 36 g 8.0 g 36 g A. Percentage Composition • Find the percentage composition of a sample that is 28 g Fe and 8.0 g O. 100 = 78% Fe %Fe = 100 = 22% O %O =
A. Percentage Composition • How many grams of copper are in a 38.0-gram sample of Cu2S? Cu2S is 79.852% Cu (38.0 g Cu2S)(0.79852) = 30.3 g Cu
36.04 g H2O 147.02 g H2O A. Percentage Composition • Find the mass percentage of water in calcium chloride dihydrate, CaCl2•2H2O? 24.51% H2O %H2O = 100 =
CH3 B. Empirical Formula • Smallest whole number ratio of atoms in a compound C2H6 reduce subscripts
B. Empirical Formula 1. Find mass (or %) of each element. 2. Find moles of each element. 3. Divide moles by the smallest # to find subscripts. 4. When necessary, multiply subscripts by 2, 3, or 4 to get whole #’s.
What ??????????How can I remember? • There is a poem to help • Percent to Mass • Mass to Mole • Divide by Small • Multiply ‘til Whole • Sometimes the mole values are still not whole numbers so multiply by the smallest factor that will make them whole numbers
1.85 mol N 1.85 mol B. Empirical Formula • Find the empirical formula for a sample of 25.9% N and 74.1% O. 25.9 g N 1 mol N 14.01 g N = 1.85 mol N = 1 N 74.1 g 1 mol 16.00 g = 4.63 mol O = 2.5 O
N2O5 B. Empirical Formula N1O2.5 Need to make the subscripts whole numbers multiply by 2
Example of Determining Empirical Formula • Ex. What is the empirical formula of a compound that is 25.9% nitrogen and 74.1% Oxygen? • 25.9g N 1 mol N = 1.85 mol N • 14.0g N • 74.1g O 1 mol O = 4.63 mol O • 16.0g O • Divide each element by the lowest number of • moles in the compound: • 1.85 mol N = 1 mol N • 1.85 • 4.63 mol O = 2.50 mol O • 1.85 • Now find the lowest common denominator. • 1 mol N x 2= N2 2.5 mol O x 2 = O5 • Therefore, N2O5 is the empirical formula
Practice Problems Calculate the empirical formula for a compound that is 94.1% O and 5.90%H Calculate the empirical formula for a compound that is 79.8% C and 20.2% H
C2H6 C. Molecular Formula • “True Formula” - the actual number of atoms in a compound CH3 empirical formula ? molecular formula
C. Molecular Formula 1. Find the empirical formula. 2. Find the empirical formula mass. 3. Divide the molecular mass by the empirical mass. 4. Multiply each subscript by the answer from step 3.
28.1 g/mol 14.03 g/mol C. Molecular Formula • The empirical formula for ethylene is CH2. Find the molecular formula if the molecular mass is 28.1 g/mol? empirical mass = 14.03 g/mol = 2.00 2(CH2) C2H4
Example of Molecular Formula Calculate the molecular formula of the compound whose Molar mass is 60.0g and empirical formula is CH4N.