Significant Digits
Significant Digits. Aluminum Foil Investigation. Your teacher will provide you with a hand out of an investigation in which you will be trying to determine the thickness of a piece of aluminum foil You will get into groups of 2 to 3 and complete the investigation. Hints for Investigation.

Significant Digits
E N D
Presentation Transcript
Aluminum Foil Investigation • Your teacher will provide you with a hand out of an investigation in which you will be trying to determine the thickness of a piece of aluminum foil • You will get into groups of 2 to 3 and complete the investigation
Hints for Investigation • Does anyone need a few hints to get them started? • Then density of aluminum is 2.70 g/cm3 • The equation for density is • Is anyone still having trouble? • Here’s another hint:
Using Measurements In Science • In the previous activity you were forced to take several measurements such as length, volume, and mass • We take measurements like this every day of our lives and often take them for granted • In science we use a standard system of measurements called the International System of Units (SI units) so that everyone is using the same system
Measurement and Uncertainty • When you were doing your calculations in the previous activity how accurate where your answers? • How accurate did you measure the aluminum with your ruler? • When working in science you need to know how much you can depend on measurements • You need to know when something was just an approximation
Measurement and Uncertainty • An example: How many great lakes are there? • There are 5 great lakes • What about the amount of millilitres in 1 L? • Number of seconds in 1 minute? • These are numbers that you can count or that are true by definition (exact numbers) • Now think about the numbers you were using in the activity, were they exact numbers?
Measurements and Uncertainty • In this course you will be using equipment such as rulers, balances and other scales to measure • You will be calculating most of your results with a calculator • These pieces of equipment as well as your ability to use them will affect your ability to record and communicate measurements and calculations
Significant Digits, Certainty, and Measurements • All measurements involve uncertainty • You cannot measure anything with complete certainty • The last (farthest right) digit in any measurement is always an estimate • Significant digits: are the digits that you record when you measure something (include digits that you are certain about and a final uncertain digit that you estimated
Example • 4.28 g has three significant digits: • The first two digits (the 4 and the 2) are certain • The last digit (the 8) is an estimate and therefore it is uncertain • The value 4.3 has two significant digits • Which digits are certain and which are uncertain?
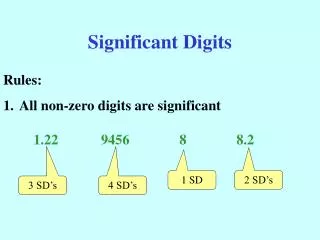
Rules for Determining Significant Digits • All non-zero numbers are significant Examples: • 7.886 has four significant digits • 19.4 has three significant digits • 527.266 992 has significant digits nine
Rules for Determining Significant Digits • Zeros placed between other digits are always significant. Examples: • 408 has three significant digits • 25 074 has significant digits • 7003 has how many significant digits? five
Rules for Determining Significant Digits • In a measurement with a decimal point, zeros placed before other digits are not significant. Examples: • 0.0907 has three significant digits. • 0.000 000 000 06 has . • 0.0056 has how many significant digits? one significant digit
Rules for Determining Significant Digits • Zeros that are located to the right of a value may or may not be significant • Zeros placed after nonzero digits after a decimal are significant Examples: • 9.100 has 4 significant digits • 802.0 hassignificant digits. • 7400.0 has how many significant digits? • If there is no decimal place then it becomes a bit more difficult to identify if the zeros to the right are significant 4
Rules for Determining Significant Digits • Zeros that are located to the right of a value may or may not be significant • If a decimal is not present, zeros to the right of the last non-zero digit are not significant. Examples: • 1200 has two significant digits • 520 has significant digits • 30 400 000 has how many significant digits? • What if you used a measuring tape to measure the length of a box and the length was exactly 200cm, how would you report this answer so that people know there are three significant digits? 2
Rules for Determining Significant Digits • Scientific notation is used to indicate whether zeroes at the end of a measurement are significant • Instead of reporting our length measurement as 200cm we would report it as 2.00xcm • When a measurement is written in scientific notation all digits in the coefficient are significant Examples: • has three significant digits • has significant digits • has how many significant digits? 5
Practice Problems (as a class) • Write the following quantities in your notebook. Beside each record the number of significant digits. • 24.7 kg • 247.7 mL • 247.701 mg • 0.247 01 L 3 significant digits 4 significant digits 6 significant digits 5 significant digits
Practice Problems (on your own) • Write the following quantities in your notebook. Beside each record the number of significant digits. • 8.9300x km • 2.5 g • 0.0003 mL • 923.2g 5 significant digits 2 significant digits 1 significant digits 4 significant digits
Homework!!! • Complete worksheet “How many significant digits for each number?” • Complete questions 1 and 2 on worksheet “Significant Digits and Rounding” BOOHOO!!!